Home » Oil and Gas » Hydraulic Fracturing Fluids
Hydraulic Fracturing Fluids - Composition and Additives
Republished from: Modern Shale Gas Development in the United States by the U.S. Department of Energy.
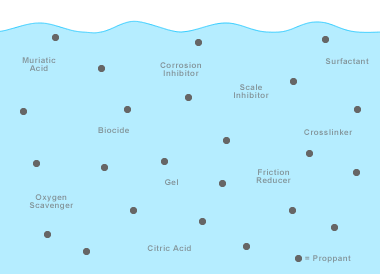
Hydraulic fracturing fluids: A wide variety of chemical additives are used in hydraulic fracturing fluids. They include: dilute acids, biocides, breakers, corrosion inhibitors, crosslinkers, friction reducers, gels, potassium chloride, oxygen scavengers, pH adjusting agents, scale inhibitors, and surfactants. These chemical additives typically might make up just 1/2 to 2 percent of the fluid. The remaining 98 to 99 1/2 percent of the fluid is water. Proppants such as sand, aluminum shot, or ceramic beads are frequently injected to hold fractures open after the pressure treatment is completed.
What is Hydraulic Fracturing?
The current practice for hydraulic fracturing treatments of shale gas reservoirs is to apply a sequenced pumping event in which millions of gallons of water-based fracturing fluids mixed with proppant materials and thickening agents are pumped in a controlled and monitored manner into the target shale formation above fracture pressure [1].
Hydraulic Fracturing Additives
The fracturing fluids used for gas shale stimulations consist primarily of water but also include a variety of additives. The number of chemical additives used in a typical fracture treatment varies depending on the conditions of the specific well being fractured.
A typical fracture treatment will use very low concentrations of between 3 and 12 additive chemicals depending on the characteristics of the water and the shale formation being fractured. Each component serves a specific, engineered purpose [2].
Friction Reducing (Slickwater) Additives
The predominant fluids currently being used for fracture treatments in the gas shale plays are water-based fracturing fluids mixed with friction-reducing additives (called slickwater) [3]. The addition of friction reducers allows fracturing fluids and proppant to be pumped to the target zone at a higher rate and reduced pressure than if water alone were used.
This video illustrates the equipment, materials and procedures used in the hydraulic fracturing process. It applies to the use of hydraulic fracturing combined with horizontal drilling in the development of a natural gas well in an organic-rich shale. It was prepared by Chesapeake Energy.
Other Additives and Proppants
In addition to friction reducers, other additives include: biocides to prevent microorganism growth and to reduce biofouling of the fractures; oxygen scavengers and other stabilizers to prevent corrosion of metal pipes; and acids that are used to remove drilling mud damage within the near-wellbore area [4]. These fluids are used not only to create the fractures in the formation but also to carry a propping agent (often silica sand or sintered bauxite) which is deposited in the induced fractures.
The make-up of fracturing fluid varies from one geologic basin or formation to another. A list of potential additives is given in Table 1 [5]. Evaluating the relative volumes of the components of a fracturing fluid reveals the relatively small volume of additives that are present. Overall the concentration of additives in most slickwater fracturing fluids is a relatively consistent 0.5% to 2% with water making up 98% to 99.5%.
Fracturing Fluids Vary from One Play to Another
Because the make-up of each fracturing fluid varies to meet the specific needs of each area, there is no one-size-fits-all formula for the volumes for each additive. In classifying fracturing fluids and their additives it is important to realize that service companies that provide these additives have developed a number of compounds with similar functional properties to be used for the same purpose in different well environments.
The difference between additive formulations may be as small as a change in concentration of a specific compound. Although the hydraulic fracturing industry may have a number of compounds that can be used in a hydraulic fracturing fluid, any single fracturing job would only use a few of the available additives. It is not uncommon for some fracturing recipes to omit some compound categories if their properties are not required for the specific application.
Most industrial processes use chemicals and almost any chemical can be hazardous in large enough quantities or if not handled properly. Even chemicals that go into our food or drinking water can be hazardous. For example, drinking water treatment plants use large quantities of chlorine. When used and handled properly, it is safe for workers and near-by residents and provides clean, safe drinking water for the community.
Although the risk is low, the potential exists for unplanned releases that could have serious effects on human health and the environment. By the same token, hydraulic fracturing uses a number of chemical additives that could be hazardous, but are safe when properly handled according to requirements and long-standing industry practices. In addition, many of these additives are common chemicals which people regularly encounter in everyday life.
| Information Sources |
|
[1] Harper, J. 2008. The Marcellus Shale - An Old "New" Gas Reservoir in Pennsylvania. Pennsylvania Geology. v 28, no 1. Spring 2008. Published by the Bureau of Topographic and Geologic Survey, Pennsylvania Department of Conservation and Natural Resources.
[2] Schlumberger. Multistage Fracturing Services. Last accessed July 2022. [3] Ketter, A.A., J. L. Daniels, J. R. Heinze, and G. Waters. A Field Study Optimizing Completion Strategies for Fracture Initiation in Barnett Shale Horizontal Wells. SPE 103232. [4] Schlumberger. PowerSTIM Well Optimization Service. Last accessed July 2022. [5] Modified from: Arthur, J.D., B. Bohm, and M. Layne. 2008. ALL Consulting. Hydraulic Fracturing Considerations for Natural Gas Wells of the Marcellus Shale. Presented at the GWPC Annual Forum in Cincinnati, OH. September 2008. [6] U.S. Department of Energy, (2009). Modern Shale Gas Development in the United States: A Primer. Work Performed Under DE-FG26-04NT15455. Prepared by The Ground Water Protection Council and ALL Consulting. |
Dilution and Neutralization of Additives
Table 1 provides a summary of the additives, their main compounds, the reason the additive is used in a hydraulic fracturing fluid, and some of the other common uses for these compounds. Hydrochloric acid (HCl) is the single largest liquid component used in a fracturing fluid aside from water; while the concentration of the acid may vary, a 15% HCl mix is a typical concentration. A 15% HCl mix is composed of 85% water and 15% acid, therefore, the volume of acid is diluted by 85% with water in its stock solution before it is pumped into the formation during a fracturing treatment.
Once the entire stage of fracturing fluid has been injected, the total volume of acid in an example fracturing fluid from the Fayetteville shale was 0.123%, which indicates the fluid had been diluted by a factor of 122 times before it is pumped into the formation. The concentration of this acid will only continue to be diluted as it is further dispersed in additional volumes of water that may be present in the subsurface. Furthermore, if this acid comes into contact with carbonate minerals in the subsurface, it would be neutralized by chemical reaction with the carbonate minerals producing water and carbon dioxide as a byproduct of the reaction.
| Table 1: Fracturing Fluid Additives |
| Additive Type | Main Compound(s) | Purpose | Common Use of Main Compound | ||||||
| Diluted Acid (15%) | Hydrochloric acid or muriatic acid | Help dissolve minerals and initiate cracks in the rock | Swimming pool chemical and cleaner | ||||||
| Biocide | Glutaraldehyde | Eliminates bacteria in the water that produce corrosive byproducts | Disinfectant; sterilize medical and dental equipment | ||||||
| Breaker | Ammonium persulfate | Allows a delayed break down of the gel polymer chains | Bleaching agent in detergent and hair cosmetics, manufacture of household plastics | ||||||
| Corrosion Inhibitor | N,n-dimethyl formamide | Prevents the corrosion of the pipe | Used in pharmaceuticals, acrylic fibers, plastics | ||||||
| Crosslinker | Borate salts | Maintains fluid viscosity as temperature increases | Laundry detergents, hand soaps, and cosmetics | ||||||
| Friction Reducer |
|
Minimizes friction between the fluid and the pipe |
|
||||||
| Gel | Guar gum or hydroxyethyl cellulose | Thickens the water in order to suspend the sand | Cosmetics, toothpaste, sauces, baked goods, ice cream | ||||||
| Iron Control | Citric acid | Prevents precipitation of metal oxides | Food additive, flavoring in food and beverages; Lemon Juice ~7% Citric Acid | ||||||
| KCI | Potassium chloride | Creates a brine carrier fluid | Low sodium table salt substitute | ||||||
| Oxygen Scavenger | Ammonium bisulfite | Removes oxygen from the water to protect the pipe from corrosion | Cosmetics, food and beverage processing, water treatment | ||||||
| pH Adjusting Agent | Sodium or potassium carbonate | Maintains the effectiveness of other components, such as crosslinkers | Washing soda, detergents, soap, water softener, glass and ceramics | ||||||
| Proppant | Silica, quartz sand | Allows the fractures to remain open so the gas can escape | Drinking water filtration, play sand, concrete, brick mortar | ||||||
| Scale Inhibitor | Ethylene glycol | Prevents scale deposits in the pipe | Automotive antifreeze, household cleansers, and deicing agent | ||||||
| Surfactant | Isopropanol | Used to increase the viscosity of the fracture fluid | Glass cleaner, antiperspirant, and hair color |
| Note: The specific compounds used in a given fracturing operation will vary depending on company preference, source water quality and site-specific characteristics of the target formation. The compounds shown above are representative of the major compounds used in hydraulic fracturing of gas shales. |
| More Oil |
 |
The Doorway to Hell |
 |
Oil Sands |
 |
Shale Gas Resources |
 |
Gifts That Rock |
 |
Horizontal Drilling |
 |
Oil and Gas Rights |
 |
Natural Gas Prices |
 |
Oil Shale |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
