Home » Minerals » Diamond » Lab-Grown Diamonds
Lab-Grown Diamonds
Also known as lab-created diamonds, man-made diamonds, synthetic diamonds.
Author: Hobart M. King, PhD, GIA Graduate Gemologist

Spectacular Color in Lab-Created Diamonds: Although lab-created diamonds have been produced since the 1950s, the early products fell far short of having the color and clarity needed for use in fine jewelry. The lab-created diamonds in the photo above represent the current "state of the art" in diamond creation and manufacturing. They have exceptional clarity, they have been cut to perfection, and their vibrant colors span the visible spectrum. These diamonds are from the Swarovski Created Diamond collection. They were debuted by Swarovski Group during Paris Haute Couture Fashion Week in January 2020. [1] This collection of colors stands as the most impressive ever seen in the lab-created diamond industry. Photograph by Swarovski Group.
What Are Lab-Grown Diamonds?
Lab-grown diamonds are diamonds that have been made by people. Just like the natural diamonds that form deep within the Earth, lab-grown diamonds are a crystalline form of carbon with a cubic (isometric) crystal structure.
Lab-grown diamonds have chemical, physical, and gemological properties that are the same as natural diamonds. However, the manufacturing processes used to produce lab-grown diamonds give them subtle features that trained gemologists and specialized instruments can use to separate lab-grown diamonds from natural diamonds.
An ability to distinguish natural diamonds from lab-grown diamonds is very important. In the gem and jewelry industry, many people have a strong preference for natural diamonds. They prefer them because they are made by nature and also because of their rarity.
As a result, natural diamonds sell for a higher price than lab-grown diamonds. However, many people gladly buy a lab-grown diamond because they can be purchased at a significant cost savings. Some people also prefer lab-grown diamonds for ethical and environmental reasons.
Table of Contents
 What Are Lab-Grown Diamonds? What Are Lab-Grown Diamonds? History of Lab-Grown Diamonds History of Lab-Grown Diamonds Separating Lab-Grown from Imitation Separating Lab-Grown from Imitation Separating Lab-Grown from Natural Separating Lab-Grown from Natural Appropriate Names for Diamonds Appropriate Names for Diamonds |

Diamond Abrasive Granules: Most lab-grown diamond produced today is used to make abrasive granules for cutting, drilling, and polishing processes. The pile of abrasive granules pictured here is about 1 centimeter across and weighs about 0.68 carat. We purchased 100 carats of 270/325 mesh (53/45 micron) diamond abrasive granules for $27.50, or 27.5 cents per carat. Is that less expensive than you would have guessed? Companies purchasing synthetic diamond abrasives in wholesale quantities will receive a much lower price.

Diamond Anvils manufactured from synthetic diamond are used in high-pressure testing and experiments. These anvils were manufactured from CVD diamond. Their bases measure between 5 and 10 millimeters in diameter and their culets between 1.5 and 3 millimeters. [2] United States Department of Energy Image.
History of Lab-Grown Diamonds
Lab-grown diamonds have been produced since the 1950s. Nearly all of the earliest lab-grown diamonds were put to industrial uses such as making abrasive granules for cutting, drilling, and polishing tools. At that time, lab-grown diamonds had color and clarity issues which made them unsuitable for use as gems.
One of the main incentives for developing methods of producing man-made diamonds was the fact that abrasive tool industries were starting to consume more diamond than could easily be supplied by mining.
The young diamond-growing industry had the potential to produce an unlimited and reliable supply of diamond abrasives if the cost of production was low enough to compete with natural diamond. This challenge was quickly achieved. Today diamond abrasive granules are produced in hundreds of factories in many parts of the world at a cost of under $1 per carat.
Within a few decades, lab-grown diamonds were being made pure enough and large enough that they could be used in a variety of high-tech applications. Lab-grown diamonds were being used as heat sinks in advanced computers; wear-resistant coatings on tools and bearings; high-durability windows; tiny anvils for high pressure experiments; specialized lenses; speaker domes; and much more.
In 1971 General Electric produced the first gem-quality lab-grown diamonds by the HPHT (high pressure / high temperature) process. The rough was in the form of small yellow crystals, often with metallic inclusions. Earth's atmosphere is 78% nitrogen, and it is the cause of yellow color in diamonds. Trace amounts of nitrogen in a diamond will produce a yellow color. Keeping it out of the diamond growing process was impossible at that time.
In 1989 the first diamonds were grown by the CVD (chemical vapor deposition) process. This process is better suited to growing gem-quality diamonds because fewer impurities are introduced into the diamond.
During the 1990s a small number of gem-quality diamonds were being produced in laboratories, but very few entered the marketplace. These diamonds were costly to produce, and manufacturers needed to reduce costs and improve quality to be competitive in the diamond jewelry market.
By 2010 the quality of lab-grown diamonds had improved significantly. A small but increasing number of lab-grown diamonds began entering the gem and jewelry market. Today, several companies are creating commercial quantities of beautiful lab-grown diamonds with spectacular clarity and color. Now, several percent of the diamonds entering the gem and jewelry marketplace are laboratory-grown.
In recent years, much work has been done to develop instruments that can be used to separate natural diamonds from lab-grown diamonds. Numerous companies are racing to develop a low-cost machine with a low error rate. Diamond identification abilities are needed for automatic sorting of large quantities of diamonds. Low-cost instruments are also needed for use in jewelry stores and pawn shops.

Diamond Lenses: A photograph of concave parabolic diamond x-ray lenses. Diamond lenses for focusing x-ray beams have been in use since the late 1990s. The lenses in this photo are about 1 millimeter across. [3]
Separating Lab-Grown from Imitation Diamonds
An ability to positively identify natural diamonds, lab-grown diamonds, and the many imitation materials is essential today for several reasons. First, significant price differences exist between these materials. Second, diamond customers are cautious and want to know exactly what they are buying. And, third, the reputation of every business that sells diamonds is at risk.
Diamond merchants who do not monitor and confirm the identity of inventory coming into their business expose themselves to the risk of financial loss, civil liability, and a loss of reputation. Misidentified merchandise and fraud can enter the supply chain at any level. A barrier for merchants is the high cost of testing equipment. Screening instruments cost thousands of dollars.
Fortunately, both lab-grown and natural diamonds are distinctly different from the many diamond imitations such as cubic zirconia and synthetic moissanite. These imitations differ from lab-grown diamonds and natural diamonds in many ways. They have a completely different chemical composition and a variety of different physical properties. Competent gemologists can easily recognize the imitations. Most jewelry professionals, with training and practice, can separate imitations from natural and synthetic diamonds with a simple testing device that can be purchased for less than $200.

Synthetic HPHT Diamonds grown in the High-Temperature Materials Laboratory of the Moscow Steel and Alloys Institute. They can be identified as HPHT diamonds because they exhibit both cubic and octahedral crystal faces. Photo by Wikipedian Lidvig14, displayed here under a Creative Commons License.
Separating Lab-Grown from Natural Diamonds
Contrary to some marketing messages, lab-grown diamonds and natural diamonds are not identical in every way. Lab-grown diamonds are produced at Earth’s surface by two different processes: HPHT (high pressure high temperature) and CVD (chemical vapor deposition).
Diamond crystals grown in these manufacturing processes inherit characteristics of the manufacturing process that can be used to tell them apart. These characteristics can also be used to separate them from natural diamonds.
The most obvious difference is the shape of the diamond crystals immediately after growth. Diamond crystals grown by the HPHT process normally exhibit cubic and octahedral faces. On the other hand, diamond crystals grown by the CVD process are normally tablular to blocky in shape and have a coating of graphite on four sides.

Magnetic Diamonds? You are looking at three tiny (two millimeter) crystals of HPHT diamond clinging to a common magnet. Many early HPHT diamonds were yellow in color and contained metallic inclusions. The yellow color was caused by traces of atmospheric nitrogen entering the growth process. The metallic inclusions were droplets of the iron-rich melt in which the diamonds were grown. Many early HPHT diamonds contained enough iron that they could be picked up with a common magnet. Over time, manufacturers developed methods to exclude most nitrogen and metallic inclusions.
Some lab-grown diamonds that have been faceted can be separated from natural diamonds by a trained gemologist using standard tools. Using a gemological microscope, the gemologist can sometimes identify inclusions or other features that are unique to HPHT-grown diamonds, CVD diamonds or natural diamonds. HPHT-grown diamond often contains inclusions of the molten metal flux in which the diamond is grown. CVD-grown diamonds often contain tiny dark pinpoint inclusions. Inclusions in HPHT and CVD diamonds were common during the early days of lab-grown diamonds, however, many diamond manufacturers have improved their methods to minimize these inclusions.
A microscope that enables observations under ultraviolet illumination can reveal fluorescent color and crystal growth patterns that are unique to HPHT and CVD production. But, most gemologists do not have this equipment. Even if the gemologist had that equipment, and the necessary training, many diamonds would not be identified with certainty. Thus, more sophisticated tools are needed.
Fortunately, several companies have invented portable screening devices that can separate natural diamonds from lab-grown diamonds. These devices correctly identify about 98% of natural diamonds and refer all other stones (lab-created diamonds, imitation materials, and the remaining 2% of natural diamonds) for additional testing. The cost of these screening devices starts at about $4000. More sophisticated devices that successfully separate natural, lab-grown, and imitation stones from one another can be purchased, but the cost for these starts at about $20,000, which is quite costly for many small businesses. But, to buy and sell with confidence, a business must invest in the knowledge of its staff and obtain the necessary equipment. Otherwise the business leaves itself open to errors and dishonesty in the supply chain.
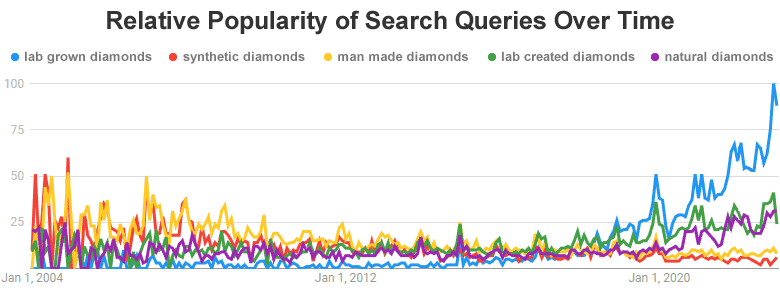
Google Trends: The image above is a screenshot from Google Trends, an online tool used to display changes in the number of people who type specific queries into Google. It can also track the relative number of people querying Google for a variety of terms. The chart above shows the relative number of people searching Google for five different queries: lab grown diamonds, synthetic diamonds, man made diamonds, lab created diamonds and natural diamonds. At the time this Google Trends chart was created (January 2023), the query "lab grown diamonds" had recently exploded from the least frequently searched into the most frequently searched of these five queries. The author believes that this surge in the use of the term "lab grown diamond" was produced by the initial, and sustained, advertising used by De Beers in the launch of their Lightbox Jewelry project. It is possible that De Beers has made a long-term mark on search query language. (Please Note: People generally do not use hyphens in search queries - you can check this for yourself on Google Trends.) If you would like to explore the current status of these search queries, please click here.

Names for Lab-Grown Diamonds: These are some of the names that people use to refer to lab-grown diamonds: Cultured, CVD/HPHT (Chemical Vapor Deposition / High-Pressure High-Temperature), Synthetic, Man-Made, LGD (Lab-Grown Diamond), and Lab-Created, among others.
| Lab-Grown Diamond Information |
|
[1] Swarovski Created Diamonds Are Debuting in Colors and Cuts Beyond Nature: a press release on the Swarovski Group website, January 21, 2020.
[2] Novel Diamond Cells for Neutron Diffraction Using Multi-Carat CVD Anvils, by R. Boehler, J.J. Molaison, and B. Haberl. Technical paper published by the United States Office of Scientific and Technical Information, August 2017.
[3] Diamond X-ray Optics: Transparent, Resilient, High-Resolution and Wavefront-Preserving, by Yuri Shvyd’ko, Vladimir Blank, and Sergey Terentyev. Technical paper published by the United States Office of Scientific and Technical Information, June 2017.
|
Appropriate Names for Diamonds
A wide variety of names have been used for diamonds that have been made by people. In the early days, the names "synthetic diamonds" and "man-made diamonds" were most often used. Synthetic diamonds was the more scientific name and the name used among technical people. Man-made diamonds was a name used by much of the general public.
Until recently the name "lab created" was used much more often than "lab grown". In December of 2013 the number of people using the name "lab created diamonds" as a Google search query was rivaling the use of "man made diamonds", which was the dominant search query for these materials at that time. In February of 2017 "lab created diamonds" became the clearly dominant query. (You can see this history for yourself in the graphic above.)
Then a major shift hit the language of Google search queries. In June of 2018, "lab grown diamonds" shot up to the leading position. This date is coincident with the first appearance of heavy online advertising for the Lightbox diamond brand of De Beers. Their ads and their website clearly used the name "lab-grown diamonds." Their product launch most likely changed the language being used by people searching Google. It also caused a major increase in the number of people who queried Google for information about diamonds made by people.
| More Diamonds |
 |
Diamond Production |
 |
Red Diamonds |
 |
US Diamond Mines |
 |
Diamonds Do Not Form From Coal |
 |
Diamond |
 |
Colored Diamonds |
 |
Canadian Diamond Mines |
 |
Brown Diamonds |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
