Home » Minerals » Diamond » Colored Diamonds
Colored Diamonds
When small imperfections create spectacular beauty
!Author: Hobart M. King, PhD, GIA Graduate Gemologist

Colored Diamonds: Diamonds can occur in a variety of beautiful colors. Images in the wonderful collection of colored diamonds shown above are used with permission of IBD Fancy Colors LLC. They include from top left, going clockwise: a heart-shaped diamond with a Fancy Vivid pink color weighing 0.70 carats; a Fancy Vivid yellowish-orange pear-shaped diamond weighing 0.85 carats; a Fancy Vivid yellow radiant cut diamond weighing 0.56 carats; a Fancy Deep brown radiant cut diamond weighing 1.00 carat; a Fancy Intense blue radiant cut diamond weighing 0.53 carats; and a Fancy Vivid green radiant cut diamond weighing 0.17 carats. They represent some of the finest hues of Fancy-color diamonds.
Table of Contents
What Are Colored Diamonds?
Colored diamonds are diamonds that have a noticeable bodycolor when viewed in the face-up position. Brown and yellow are the most common colors in natural colored diamonds. Diamonds with natural pink, blue, orange, green, red, and violet bodycolors are extremely rare.
Out of 100,000 diamonds, only a few will have one of the extremely rare colors. The color can have any intensity between "very light" and "vivid". To be considered a "colored diamond" the color must be noticeable when the diamond is in the face-up position. Black, gray, and white diamonds are also possible.

Argyle Isla: The Argyle Isla is a 1.14-carat Fancy Red radiant-cut diamond mined from the Argyle Mine in Western Australia. It is one of the most valuable diamonds in the world on the basis of dollars per carat. It was part of the Argyle Tender Heroes sale in 2017. Image Copyright 2017 by Rio Tinto.
Prices of Colored Diamonds
A combination of rarity, beauty, and intense buyer interest drives the prices of colored diamonds. The rarest colored diamonds often sell for millions of dollars per carat. They have more value for their size than almost any other material. At the same time, some colored diamonds sell for prices that many people can afford.
The most affordable colored diamonds have a brown, gray, or black color. Among colored diamonds, these colors are the least rare and most affordable. Attractive diamonds in these colors often sell for a few thousand dollars per carat. They sometimes sell for prices that are lower than diamonds on the D-to-Z color scale that are of similar size, cut, and clarity.
Yellow diamonds are next in rarity. They are more rare than brown, gray, and black diamonds. Many people consider yellow diamonds to be the most beautiful yellow gem because of their brightness, fire, and exceptional luster. Beautiful diamonds with a Fancy yellow color can often be purchased for $5,000 to $10,000 per carat in sizes under two carats. Many people do not realize that yellow diamonds are an option in this price range. Here is their chance to get a colored diamond at a surprising price.
Blue, green, and pink diamonds are much more rare than yellow diamonds. Another step up in rarity are orange, violet, and red diamonds. These colors often sell for over $100,000 per carat, and those of finest color and quality will sell for over $1,000,000 per carat.
The price of a colored diamond depends on more than its hue. Color saturation and the uniformity of color across the face of the gem are also important. Those with a pure hue are generally more desirable than those with modifying colors. As with all diamonds, the size of the gem and the quality of the cut are also very important.
In the past decade, colored diamonds grown in laboratories and natural diamonds with color produced by treatment have become more common in the market. These diamonds are not rare, and they sell for lower prices than natural diamonds with colors of natural origin. To ensure the identity of what you are purchasing, gemological laboratories can examine a diamond to determine if it is natural or laboratory grown. The labs can also determine the origin of a diamond's color. These laboratory services have a cost that is very low compared to the cost of a diamond. Identification and grading by a reputable laboratory is an essential part of any colored diamond transaction.

An artist's sketch of a collection of rough diamonds of various colors and crystal habits once owned by Gardner F. Williams, general manager of De Beers Consolidated Mines, LTD., from 1887 until 1905. The collection above nicely exhibits the colors and crystal habits of diamond. Williams was the author of The Diamond Mines of South Africa; Some Account of their Rise and Development (1905) [1]. At the time that this collection was assembled, colored diamonds were a novelty that obviously attracted the attention of Williams; however, the marketplace did not celebrate their true rarity and price them anywhere near what they command today.
Common Causes of Color in Diamond |
|
| Pink | Pink is a rare natural color in diamonds. It occurs when the diamond is subjected to stress within the Earth, and those forces cause glide planes of carbon atom displacement within the diamond crystal. When light passes through the planes, red light is selectively transmitted. The red light appears pink when the selective transmission is weak. The selective transmission is rarely strong enough to produce a red color. |
| Red | Red diamonds are extremely rare in nature, and they are the most valuable diamonds when in a pure hue. Like pink diamonds, they have been subjected to stress which deformed the diamond crystal lattice, causing glide planes of carbon atom displacement. When light passes through the planes of displacement, the red wavelengths of light are selectively transmitted. Weak transmission of red light will produce a pink diamond. |
| Orange | Orange diamonds are very rare. The defect(s) that produce the orange color have not been determined with certainty and may vary from one orange diamond to another. The defects in orange diamonds cause them to selectively absorb blue light and selectively transmit orange. |
| Yellow | Yellow is the second most common natural color in diamonds. The color is usually caused by nitrogen atoms substituting for carbon in the diamond crystal lattice. This defect causes diamond to selectively absorb blue light and selectively transmit yellow. |
| Green | Green diamonds are very rare in nature. The color usually develops when high-energy radiation emitted by nearby radioactive mineral grains penetrates the diamond. The radiation knocks carbon atoms out of their position in the diamond crystal lattice, and that defect causes the diamond crystal to selectively absorb red light and selectively transmit green. Green color can also be a result of defects produced by the presence of nitrogen, hydrogen, or nickel within the diamond crystal. |
| Blue | Blue diamonds are rare in nature. The blue color is most often caused by boron atoms substituting for carbon atoms in the crystal lattice of diamonds that have formed at extreme depths. As little as one boron atom per million carbon atoms can produce a noticeable blue color. Boron in the diamond crystal causes the selective absorption of red light and the selective transmission of blue. [2] |
| Violet | Violet is one of the rarest natural colors in diamond. It is sometimes caused by substitution of hydrogen in place of carbon in the diamond crystal lattice. |
| Purple | Purple is another rare color in diamond. In a study of 50 purple diamonds by GIA, they often found H3 and N3 color centers, sufficient enough to influence color. Purple diamonds and some pink diamonds modified by purple often exhibit color concentration along glide planes of carbon atom displacement. [4] |
| Brown | Brown is the most common natural color in diamonds. The color develops when plastic deformation creates planes of missing and displaced carbon atoms in the diamond crystal lattice. These are known as glide planes, and they are where the brown color is concentrated. They can appear as a series of parallel color bands in the diamond known as "graining". |
| White | White diamonds occur in nature when the diamond has dense clouds of fine, reflective inclusions. The numerous inclusions can interfere with the passage of light and give the diamond a translucent or opalescent appearance. |
| Black | Black diamonds with a natural color usually contain such a high density of mineral inclusions that very little light passes through the gem. Common inclusions in black diamonds include graphite, pyrite, or hematite. Black color in heavily fractured diamonds can be caused by graphitization of the fracture surfaces. |
| Please note: The causes of color listed above are just a few of the many causes of color in natural diamonds. Numerous other natural defects can produce color. People also change or induce color in diamonds by treatments that include irradiation, heating, and coating - and by combinations of multiple treatments. There are many causes of color in diamonds, and researchers are only beginning to understand them. | |
Who Buys Colored Diamonds?
Colored diamonds are among the most beautiful and eye-catching gems. Many colored diamonds are purchased by consumers. They buy them already set in jewelry or have them set into specially designed jewelry that they intend to wear. These people love beautiful gems, and a custom-designed setting with a sparkling colored diamond is the ultimate jewelry item.
Museums are another buyer of colored diamonds. They purchase colored diamonds when building or improving their gemstone and mineral exhibits. Colored diamonds are among the most popular exhibits at many museums.
Gem collectors purchase a lot of colored diamonds. In fact, many museums have received their colored diamonds through the gifts and bequests of gem collectors.
Research institutions buy colored diamonds for study and for their reference collections. The Gemological Institute of America has a large collection of colored diamonds that they use for study and to improve their ability to grade colored diamonds. They maintain the world's largest masterstone sets for color-grading colored diamonds.
A few people purchase colored diamonds in hopes of price appreciation or to own a very small item with a very high value. Colored diamond prices have climbed steadily for the past several decades as more people have learned about them, and public interest has been fueled by spectacular prices seen in public auctions. However, there is no guarantee that this price trend will continue. Anyone interested in investing in colored diamonds would be very wise to learn a lot about them and find a trusted seller who can offer them at investor prices.
Le Vian, a jewelry design and manufacturing company, has trademarked the name "Chocolate Diamonds". They source brown diamonds within their "chocolate color range" and use them to produce a world-famous brand of diamond jewelry. Their "chocolate diamonds" are budget-priced and easy to find in stores. They make the purchase of a colored diamond accessible to people who might otherwise not be able to afford them.
Causes of Color In Diamonds
Natural diamond is a mineral and a rare form of crystalline carbon. A diamond composed entirely of carbon and with a perfect crystalline structure will be colorless. Diamonds that perfect rarely exist. Instead, most diamond crystals accumulate defects during crystal growth and during their long history within the Earth. Multiple types of defects can be incorporated in a single diamond.
Some of these defects can modify the way that light passes through. They can cause the diamond crystal to selectively transmit some wavelengths of light and selectively absorb other wavelengths of light. Because each wavelength of light corresponds to a different color, selective transmission and absorption will determine the diamond's apparent color in the eye of the observer.
Examples of these defects include: 1) atoms of elements such as nitrogen or boron substituting for a small number of carbon atoms in the diamond crystal lattice; 2) vacant sites for carbon atoms in the diamond crystal lattice; 3) minute deformations in the diamond crystal lattice; and, 4) tiny particles of non-diamond materials included in the diamond crystal. Each of these is described below in much more detail.
Although the name "defect" is used for these color-causing irregularities within a diamond crystal, the impact of their presence is limited to a color change. Buyers of colored diamonds should not be concerned about them being a durability issue.
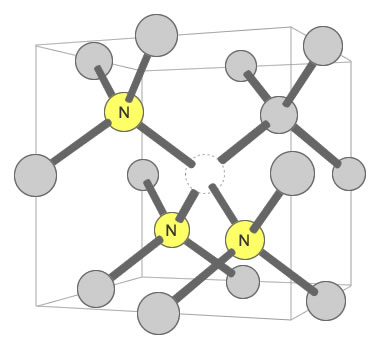
Three Nitrogens and a Vacancy Defect: Yellow color in mined diamonds can be caused by the N3 defect. It consists of three nitrogen atoms substituting for carbon atoms that surround one vacant carbon position. This defect is often accompanied by an N2 defect, and their pairing may contribute to yellow color. Illustration modified after a Creative Commons image by Materialscientist.
Atom Substitution Defects
One of the most common color-causing defects in diamond occurs when an atom of an element other than carbon substitutes for a carbon atom in the diamond crystal lattice. Substitution of nitrogen for carbon can produce a yellow diamond.
Substitution of nitrogen can cause the diamond crystal to selectively absorb blue wavelengths of light and selectively transmit yellow. That transmitted yellow light reaches the observer's eye and causes the observer to perceive an apparent yellow color in the diamond.
Out of all the different elements, only a few have atoms tiny enough to substitute for carbon in a way that produces color. Nitrogen, boron, and hydrogen are among the few that have the ability to substitute naturally for carbon in the diamond crystal lattice.
Substitution of boron for carbon can cause a diamond to appear blue. Substitution of hydrogen can cause a violet color in some diamonds.
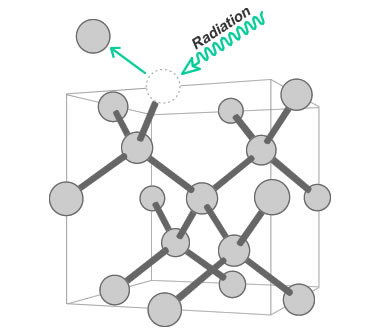
Radiation-Induced Defect: Color in green diamonds is often a result of radiation exposure. The radiation knocks carbon atoms out of their position in the diamond crystal lattice. Illustration modified after a Creative Commons image by Materialscientist.
Vacancy Defects
Exposure to radiation can contribute to the color of a diamond. If a diamond is located near radioactive mineral grains during its time within the Earth, it can be exposed to a stream of high-velocity particles. These high-velocity particles can knock carbon atoms out of their lattice position within the diamond.
This vacancy defect can cause the selective absorption of red light and the selective transmission of green. When the transmitted green light reaches the observer's eye, the diamond will appear green. This is the cause of color in many naturally green diamonds.

Pink Graining in Diamond: In this photomicrograph, you are looking into the interior of a rough diamond through a small polished window on its surface. The pink vertical lines are "graining" caused by plastic deformation of the diamond crystal lattice. Each line traces a slip plane within the diamond where carbon atoms have been displaced. In this view the slip planes intersect the polished window at a right angle. Each slip plane is a defect in the diamond that causes the diamond to selectively absorb green light and selectively transmit red. Note the tiny offsets where the slip planes intersect the edges of the polished window. A small amount of pink graining in a diamond crystal can cause that diamond crystal to have a pink color. Where the amount of graining is very high the diamond crystal can have a red color. Pink and red diamonds are caused by the same type of defect. The color (pink or red) is determined by the abundance of pink graining in the stone. Photograph by the United States Naval Research Laboratory. [5]
Crystal Lattice Deformation
Diamonds form and spend millions of years in the deep Earth environment where they are exposed to extreme temperatures and pressures. Some diamonds also spend part of their history in areas of Earth's interior that are subjected to the lateral forces of plate tectonics.
Although diamond is an extremely durable material, the forces of Earth's interior can cause the carbon atoms in a diamond's crystal lattice to slip slightly out of alignment. This movement usually occurs across surfaces within the diamond known as "glide planes" (in the gemological literature these features are also called "slip planes" or "lamellae").
These glide planes are not "fractures". They are planes within the diamond along which a tiny shift of the carbon atoms has taken place - without damaging the structural integrity of the diamond. However, the glide planes do alter the way that light passes through the diamond crystal. They selectively absorb some wavelengths (colors) of light passing through the diamond and selectively transmit other wavelengths (colors).
When glide planes are observed through a microscope, a small amount of color can often be perceived (see the accompanying photo). Some diamonds have multiple parallel glide planes, and that can increase the intensity of the diamond's color. The color patterns and slight relief produced by the glide planes often have an appearance similar to wood grain. For that reason, some gemologists use the term "graining" when referring to these features. Most graining displays as brown in color, and in very rare instances, as pink or red.
Occasionally the graining is strong enough to be visible with the unaided eye. When visible at 10x magnification or less, the graining can be considered a clarity characteristic. Graining visible in diamond at 10x magnification can influence both the bodycolor of the gem and its clarity grade. If you would like to see photomicrographs of glide planes in numerous diamonds, see the article about Natural-Color Pink, Purple, Red, and Brown Diamonds in the Winter 2018 issue of Gems and Gemology. [6]
The Argyle mine in Australia is the best-known mine in the world for producing diamonds colored by graining defects. About 80% of the diamonds produced at the mine are brown in color. Argyle is also the world's most important source of diamonds with a pink or red natural color. The mine usually produces a few hundred carats of pink diamonds per year. However, in an entire decade the mine will produce only a few dozen carats of diamonds with a pure red hue.
Mineral Inclusions
Black color in diamonds can be caused by a high density of inclusions - so many inclusions that they interfere with the passage of light. The inclusions in black diamonds are usually opaque minerals such as graphite, hematite, or pyrite. When the inclusions have a very small particle size and are uniformly distributed, they can render the diamond opaque, with a handsome black color and a highly reflective surface. Some black diamonds obtain their color from small surface-reaching fractures that have been graphitized.
The name "white diamonds" is often used in reference to transparent diamonds on GIA's D-to-Z color grading scale. That usage is common, but it is not precise.
The name "white diamonds" is best used for diamonds with a white bodycolor. White diamonds often contain dense clouds of very fine-grained transparent to translucent mineral inclusions that interfere with the passage of light through the diamond. These inclusions can cause the diamond to have a white translucent color, and if they are slightly reflective they can produce an opalescent "glow" within the gem. These diamonds have a truly white bodycolor and are considered to be "colored diamonds" or "Fancy white" diamonds if the quality of their white color merits these designations.

Fancy Vivid Orangy Yellow: In 2018, ALROSA surprised the diamond market by auctioning a collection of 250 colored diamonds in Hong Kong. Known as the "True Colors" auction, ALROSA intends to make the sale an annual event and reports that they will easily be able to support the annual sale because they produce at least 7000 carats of colored diamonds per year. The stone above is an oval-cut, 15.11-carat, Fancy Vivid orangy yellow, VVS2 clarity gem. Photograph by ALROSA.
Modified and Secondary Hues
Very few diamonds have a pure hue, such as red, blue, green, orange, or yellow. These are the most sought-after diamonds, and their prices are usually very high. Instead, most colored diamonds have a color that is intermediate between pure hues such as yellow and green. As an example, yellow diamonds might exhibit hints of green (greenish yellow) or orange (orangy yellow). The possible colors of diamond form a gradient of colors across the visible spectrum.
The existence of all of these intermediate colors suggests that the processes that cause color in diamonds are very complex, and that multiple causes of color can exist in a single diamond. This is what produces the incredible spectrum of diamond colors that are possible in nature.
The diamonds that depart from a pure hue provide buying opportunities to the shopper. They usually sell for lower prices than diamonds with a pure hue. Buyers who like them and can afford them can obtain a colored diamond at a lower price than a diamond of premium color.

The Hope Diamond is the world's most famous blue diamond. It began as a deep blue rough diamond mined in the Golconda Sultinate in the 1600s. It was first cut into a gem known as the "Tavernier Blue"; recut to yield a stone named the "French Blue"; and was finally cut into a diamond that was eventually named the "Hope Diamond". It weighs 45.52 carats and has a Fancy Dark grayish blue color. It is on display at the National Museum of Natural History in Washington, D.C. Photograph from the archives of the Smithsonian Institution.
Sources of Colored Diamonds
Mines of the Golconda Sultanate, India
One of the all-time greatest sources of diamonds, and the source of many of the world's most famous colored diamonds, are the diamond mines of the Golconda Sultanate (today the Indian states of Telangana and Andhra Pradesh). Diamond mining began in the Golconda Sultanate as early as the 1400s and continued well into the 1600s.
These mines were the source of many important colored diamonds including the Nassak (43.8 carats, blue), Sancy (55.23 carats, yellow), Daria-i-Noor (182 carats, pink), Hope (67 carats, blue), Dresden Green (41 carats, green), Princie (34.65 carats, pink), Wittelsbach-Graff (31 carats, blue), Idol's Eye (70.21 carats, blue), Agra (31.41 carats, pink), and Noor-ul-Ain (60 carats, pink).

Argyle Liberte: The Argyle Liberte is a 0.91-carat Fancy Deep gray-violet radiant-cut diamond mined from the Argyle Mine in Western Australia. It was part of the 2017 Argyle Tender sale. Image Copyright 2017 by Rio Tinto.
The Argyle Mine, Western Australia
The world's most important source of colored diamonds since 1985 has been the Argyle mine of Western Australia. It has held the distinction of being a reliable source of pink, red and violet diamonds - which can command prices of over $1 million per carat.
Originally sent to be crushed for abrasive granules, the beauty of Argyle's brown diamonds was not fully appreciated until the 1980s. On the lower end of the colored diamond price range, Argyle's brown diamonds made colored diamonds affordable to almost anyone. Levian has best taken advantage of these inexpensive colored diamonds by creating their trademarked "chocolate diamonds", which they often set in "strawberry gold". Unfortunately, the Argyle Mine ceased production in November of 2020.

ALROSA's True Colors: One of the online advertisements used to promote ALROSA's 2019 True Colors Auction, in which they offered 200 colored diamonds with GIA reports, with a total weight of 2052.88 carats. The auction sale was held at the Hong Kong Jewellery & Gem Fair on September 16 to 20, 2019. Example diamonds from the sale are shown on this page.
The Mines of ALROSA, Russia
ALROSA, a Russian diamond-mining conglomerate, has numerous mines where colored diamonds are found. In 2018, ALROSA held their first "True Colors" auction, where they offered over 200 fancy colored diamonds of various shapes and colors. ALROSA announced that they intend to "become the leader of the fancy color diamonds market" and become "the largest producer of fancy color diamonds by volume". [7]
Almazy Anabara, a subsidiary of ALROSA, produces colored diamonds from several mines in the Sakha Republic of northeastern Russia. All of these mines work placer deposits along the Ebelyakh River, north of the Arctic Circle. There they have found spectacular yellow, pink, lilac, and blue diamonds in these alluvial deposits. The company is optimistic that they will find a rich primary source(s) for these placers. [8]
ALROSA's Lomonosov deposit has produced numerous yellow, pink, purple, blue and yellowish green diamonds. Like the Argyle Mine of Western Australia, the Lomonosov deposit is located within an orogenic region where the rocks have been subjected to tectonic processes. This is believed to contribute to the presence of color in many Lomonosov diamonds. [8]
Sources by Color
No mine can be considered an abundant source of premium-color pink, red, orange, blue, green, or violet diamonds. These colors are truly rare. However, a few mines are known to regularly produce small quantities of colored diamonds. Some of the better-known sources are summarized below by color.

Fancy Vivid Purplish Pink: The photo above is of a Fancy Vivid purplish pink diamond, part of ALROSA's 2019 True Colors auction at the Hong Kong Jewellery & Gem Fair. This is a spectacular diamond, weighing 0.55 carat, with a GIA cut grade of excellent, and a clarity grade of internally flawless. It was found at ALROSA's Arkhangelsk mine. With the Argyle Mine in Australia now closed, ALROSA could become the world's premier producer of colored diamonds. Photograph by ALROSA.
Sources of Pink Diamonds
The Argyle Mine in Australia is currently a source for small quantities of pink diamonds. The Golconda mines of India have been historic sources of pink diamonds. Occasional pink diamonds are produced at the Lulo alluvial project in Angola, the Minas Gerais area of Brazil, the Williamson mine in Tanzania, and the Premier mine in South Africa. In Russia, the Lomonosov, Yakutia, and Arkhangelsk Mines owned by ALROSA have been producing a number of pink diamonds every year.
Sources of Red Diamonds
Since 1985, the Argyle Mine in Western Australia has been the source of almost all of the red diamonds that have entered the market. But, three of the world's most famous red diamonds came from other sources. The Hancock Red (which sold for $926,315 per carat in 1987) was found in Brazil. The Moussaieff Red, the largest red diamond known, was also found in Brazil. And, the DeYoung Red, the third-largest red diamond known, was found at an unknown source.
Sources of Orange Diamonds
Orange diamonds are extremely rare, and no locality has established a name as a steady source.
Sources of Yellow Diamonds
Yellow is the second most common diamond color after brown. Diamonds with Fancy-grade yellow color are found in small quantities at many mines throughout the world.
Sources of Green Diamonds
The Golconda mines in India have been a historic source for a few green diamonds. Small numbers of green diamonds have also been mined in Brazil, the Central African Republic, Guyana, Russia, South Africa, and Zimbabwe.

The Letlapa Tala Collection: These five rough blue diamonds were all found during a single week in September 2020 at the Cullinan Mine, owned by Petra Diamonds. They weigh 25.75, 21.25, 17.57, 11.42, and 9.61 carats. The Cullinan Mine is the world's most important source of blue diamonds. Photograph by Petra Diamonds.
Sources of Blue Diamonds
The Cullinan Mine (formerly the Premier Mine) in South Africa is the best-known producer of blue diamonds. Since 2009, Petra Diamonds, owner of the Cullinan Mine, has auctioned several large blue diamonds weighing over 25 carats.
The accompanying image displays the Letlapa Tala Collection, a group of five rough Type II blue diamonds, all found at the Premier Mine during a single week in September 2020. "Letlapa Tala" means "blue rock" in Northern Sotho, the predominant language of the Cullinan area. [9] The Cullinan Mine has produced the world's largest rough diamond, the world's largest faceted diamond, and a significant share of the world's diamonds weighing over 100 carats.
In Russia, small numbers of blue diamonds have been found ALROSA's placer deposits in the Sakha Republic, and at their Lomonosov deposits in the Arkhangelsk region. Other mines in Brazil, India, Indonesia, Russia, Sierra Leone, and South Africa have produced occasional blue diamonds.
Sources of Violet Diamonds
The Argyle Mine of Western Australia has been the most persistent source of diamonds with a pure violet hue. However, do not allow that statement to make you think that violet diamonds are abundant - because over the entire history of the mine, less than 100 carats of rough violet diamonds have been found there. Argyle might be considered as "the most persistent source" because a few carats have been found in each of several years, and rarely are diamonds with a pure violet hue found anywhere else.
Sources of Purple Diamonds
ALROSA has been a recurring source for small numbers of purple diamonds. They are occasionally found in all of the company's Siberian deposits and most often found in pipes of the Mir kimberlite field. [4]
ALROSA is also finding lilac (pinkish-purple) diamonds in its Almazy Anabara placer deposits along the Ebelyakh River. In 2017, a 20.18-carat octahedral crystal with a lilac color was found there. Lilac diamonds are also found in small numbers in ALROSA's Lomonosov deposit. [8]
Sources of Brown Diamonds
Brown is the most common color of colored diamonds. Brown diamonds are abundant and found in diamond deposits throughout the world. The best-known mine for producing brown diamonds is the Argyle mine in Australia, where over 80% of the gem-quality diamonds produced are brown. A few Alrosa mines in Russia produce significant quantities of brown diamonds.
Colored Diamond Treatments
Information in the article above is concerned with the causes of natural color in diamonds. However, a large and growing number of treatments are being used to modify the color of diamonds. These treatments can be divided into two groups: 1) surface treatments that modify the apparent color of the diamond; and, 2) crystal lattice modifications that change the way that light travels through the diamond.
1) Surface Treatments involve applying a substance to the surface of a diamond that alters the color of light that travels through the diamond, or, applying a substance to the surface of a diamond that alters the color of light that is reflected from the surface of a diamond.
Some of the earliest and simplest treatments involved applying ink or paint to the culet, pavilion, or girdle of a diamond. The application of a colored material to these surfaces of a diamond can cause that color to be reflected through the diamond, changing the apparent color of the diamond when it is viewed in the face-up position. These changes are not permanent, but if they are applied to surfaces that are protected by a setting that prevents abrasion or removal, they can persist for a long time.
Another type of treatment involves applying metal oxide coatings to the surface of a diamond. These colored coatings reflected color from the surface of the diamond and reflect color through the interior of the diamond. The coating, instead of the diamond, is responsible for the color of light that reaches the eye of the observer. Examples are: SiO2 coatings doped with gold to produce pink color; SiO2 coatings doped with silver to produce blue or yellow color; and Fe2O3 coatings to produce orange color. These treatments are not permanent but can produce a vivid apparent color. [10]
2) Crystal Lattice Modifications are done using irradiation, high-temperature / high-pressure annealing, low-pressure / high-temperature annealing, and combinations of treatments applied in different sequences. These treatments produce the same types of defects found in natural diamonds that have been exposed to a similar history of conditions within the Earth. The results of these treatments vary and can be dependent upon the characteristics of the original diamond.
Modification of a diamond's color by any type of treatment can be a legitimate business practice if that treatment is fully disclosed to, and understood by, people who purchase the diamond. Any special care requirements must also be disclosed at the same time. Diamonds with a color produced by treatment should be sold for lower prices than similar-color diamonds with a color of natural origin.
Many buyers gladly purchase diamonds that have treatment-produced color and enjoy their significantly lower prices. At the same time, many buyers strongly dislike gems that have been treated because they are only interested in diamonds with natural color. These buyers insist upon full disclosure of any treatments and are willing to pay a premium price for natural-color diamonds.
A brief introduction to diamond treatments and information about the range of colors possible can be found in a GIA article here. [11]
| Colored Diamond References |
|
[1] The Diamond Mines of South Africa: Some Account of their Rise and Development, by Gardner F. Williams; The Macmillan Company; 1902, Volume 2, frontispiece. [2] NOVA: Treasures of the Earth: DVD by Doug Hamilton and others; a NOVA / PBS program, 180 minutes, 2016. [3] The Nature of Diamonds: by George E. Harlow; a book published by Cambridge University Press in association with the American Museum of Natural History; 278 pages; 1998. [4] Natural-Color Purple Diamonds from Siberia: by Sergey V.Titkov, James E. Shigley, Christopher Breeding, Rimma M. Mineeva, Nikolay G. Zudin and Aleksandr M. Sergeev; Gems & Gemology, Volume 44, Number 1, pages 56 to 64, Spring 2008. [5] Naval Research Laboratory Takes a Close Look at Unique Diamonds: by Donna McKinney of the United States Naval Research Laboratory; republished on Phys.org, March 2010. [6] Natural-Color Pink, Purple, Red, and Brown Diamonds: Band of Many Colors: by Sally Eaton-Magana, Troy Ardon, Karen V. Smit, Christopher M. Breeding, and James E. Shigley; Gems & Gemology, Volume 54, Number 4, pages 352 to 377, Winter 2018. [7] Diamond Origin Is Important to Industry and Consumers Alike: sponsored content by ALROSA, published on the JCK website, last accessed July 2022. [8] Wonder Mines: an article in ALROSA Magazine (an in-house publication of ALROSA), September 2018 issue, pages 18 to 23. Hosted on the Internet Archive. [9] Petra Introduces The Letlapa Tala Collection: a press release from Petra Diamonds Limited, published October 21, 2020. [10] Serenity Coated Colored Diamonds: Detection and Durability: by Andy H. Shen, Wuyi Wang, Matthew S. Hall, Steven Novak, Shane F. McClure, James E. Shigley, and Thomas M. Moses; Gems and Gemology, Volume 43, Number 1, pages 16 to 34, 2007. [11] Changing a Diamond's Color; article on the Gemological Institute of America website, last accessed July 2022. [12] Swarovski Created Diamonds Are Debuting in Colors and Cuts Beyond Nature: a press release from the Swarovski Group, January 21, 2020. Archived copy. [13] De Beers' Stephen Lussier on Forevermark and Lightbox by Rob Bates; an article on the JCK website, published May 31, 2019. [14] The Lightbox Lab: An information page on the Lightbox website, last accessed July 2022. [15] Natural-Color Green Diamonds: A Beautiful Conundrum: by Christopher M. Breeding, Sally Eaton-Magana, and James E. Shigley; Gems & Gemology, Volume 54, Number 1, pages 2 to 27, Spring 2018. [16] Secrets of the Gem Trade: The Connoisseur's Guide to Precious Gemstones: by Richard W. Wise; a book published by Brunswick House; second; 404 pages; December 2016. [17] Diamonds and Color: Gemological Institute of America; Chapter 12; Diamonds & Diamond Grading course materials; page 13; Revised 2014 edition. [18] The Beauty of Blue Diamonds; article on the Gemological Institute of America website, last accessed July 2022. [19] Natural-Color Blue, Gray, and Violet Diamonds: Allure of the Deep: by Sally Eaton-Magana, Christopher M. Breeding, and James E. Shigley; Gems and Gemology, Volume 54, Number 2, pages 112 to 131; Summer 2018. [20] Black Diamonds: What You Need to Know; article on the Gemological Institute of America website, last accessed November 2020. [21] Characterization and Grading of Natural-Color Pink Diamonds: by John M. King, James E. Shigley, Scott S. Guhin, Thomas H. Gelb, and Matthew Hall; Gems & Gemology, Volume 38, Number 2, pages 128 to 147, Summer 2002. [22] How They're Made - Laboratory-Grown Diamonds: article on the Lightbox Jewelry website, last accessed July 2022. |
Colored Diamond Reports
If you are buying or selling a colored diamond, it is a good idea to have an independent laboratory examine the gem and determine: A) if the material is indeed diamond; B) if the diamond is natural or lab-created; C) if the diamond's color is natural or a product of treatment; D) if the diamond has been subjected to any other treatments; and, E) the color grade of the diamond.
In terms of color, the "ultimate diamond" has either a complete lack of color or a highly saturated color in a pure hue. An extremely rare diamond will occupy one of these positions.
Those which approach a complete lack of color are graded on the D-to-Z color scale developed by the Gemological Institute of America. A diamond with an absence of color earns a grade of "D" and is said to be "colorless". Progressing down the scale below D, at grades of E, F, G, etc. diamonds will exhibit very small amounts of color. The color is usually yellow, brown or gray. This grading is done in the table-down position.
A rare percentage of diamonds will exhibit traces of other colors such as pink, blue, orange, green, etc. If the color of these diamonds is noticeable in the face-up position, they will earn the term "colored diamonds" with colors described as "faint', "very light" or "light". People familiar with colored diamonds will immediately recognize "faint" color. But, inexperienced persons might not notice "faint" color unless they receive a cue to look for it or have a comparison stone nearby. Diamonds with "very light" or "light" color are much more noticeable.
Diamonds that have enough color to exceed "Z" on the D-to-Z scale will receive a grade known as "Fancy". Depending upon their tone and saturation when viewed in the face-up position, they will be given Fancy grades as follows:
 Fancy Light Fancy Light Fancy Fancy Fancy Dark Fancy Dark Fancy Deep Fancy Deep Fancy Intense Fancy Intense Fancy Vivid Fancy Vivid |
The names of these grades are important, and they are to be capitalized in all written communications. Diamonds in all of these grades can be beautiful, but those earning grades of Fancy Intense and Fancy Vivid are the most valuable. All of these diamonds have very rare color.
Grades are assigned to colored diamonds by highly trained experts who work with the aid of colored master stones for consistency. Expert grading is very important because the color grade of a diamond can make an enormous difference in its value.
Anyone who purchases a valuable diamond is wise to pay for the service of a colored diamond report. The cost of the report is very small when compared to the cost of the diamond. The best-known source of colored diamond grading reports is the Gemological Institute of America. Descriptions and fees for GIA reports can be seen here.

Color in Swarovski Created Diamonds: The biggest diamond surprise of 2020 might be the 16 spectacular colors of lab-grown diamonds debuted by Swarovski Group during Paris Haute Couture Fashion Week. This collection of colors stands as the most impressive ever seen in the lab-grown diamond industry. Photograph by Swarovski Group.
Color In Lab-Grown Diamonds
Many of the earliest lab-grown diamonds were yellow in color. Earth's near-surface atmosphere is 78% nitrogen, and keeping nitrogen out of the growing process was extremely difficult. Eventually this problem was solved, and now growers have almost complete control over the diamond-growing process. They have many options for modifying diamond color using post-growth treatments.
"As Grown" Color
Today, lab-grown diamonds with an intentional or "as grown" color are common products in the gem and jewelry industry. These diamonds are grown in environments that introduce color-causing atoms or other defects into the diamond while the crystal lattice is developing.
"Post-Growth" Treatments
Lab-grown diamonds are also subjected to "post-growth" treatments to modify their colors. Treatments include: irradiation, high-temperature / high-pressure annealing, low-pressure / high-temperature annealing, and combinations of treatments applied in different sequences. These lab-grown colored diamonds are sold in a spectrum of colors and sell for prices that are significantly less than mined diamonds with natural color - sometimes less than 1% of the cost of a similar-quality diamond with a natural color.
Lab-grown diamonds give people who are unable to afford (or who prefer not to pay) the high cost of a natural diamond with natural color an opportunity to obtain a similar-looking piece of jewelry at a much lower cost.
The Lab-Grown Color Range
In January, 2020 Swarovski Group surprised everyone by debuting their Swarovski Created Diamonds in 16 vibrant colors. They have produced the most spectacular range of hues in the lab-created diamond market today - all cut to the company's standards of precision and excellence. Swarovski plans to offer a standard selection of lab-created colored diamonds in weights ranging from 0.25 carat to 1.50 carat. [12]
The color range of the Swarovski collection spans the visible spectrum. The colors are uniform throughout the stones. The saturations are strong enough to be eye-catching and highly desirable - even in a small stone. See accompanying photo above.
Lightbox Advertising: This is one of the early online advertisements promoting the sale of Lightbox Jewelry. Note the use of the words "lab-grown diamonds" to clearly communicate the man-made origin of their diamonds, which were available in pink, blue and "white". We started seeing Lightbox Jewelry ads of various designs months before their products were available for purchase, and continued seeing them heavily through Christmas 2018 and Valentine's Day 2019. We saw some of these ads on Geology.com and other websites that we visit relevant to gems and jewelry. But, after visiting the Lightbox Jewelry website, we suddenly started seeing Lightbox ads at a high rate of frequency on many other websites that we visited, regardless of their content topic. The ads were targeted to visitor behavior rather than being space purchased on various websites. We receive no compensation for displaying the ad above, and have no agreements or relationship with LightboxJewelry.com.
The author believes that Lightbox publicity and advertising was responsible for triggering use of the term "lab-grown diamonds" by many people who use Google search. Why? One week after the De Beers announcement, the query "lab grown diamonds" exploded in relative frequency on Google Trends relative to the queries "lab-created diamonds", "man-made diamonds". We explain that in greater detail in our article about lab-grown diamonds. If you want to see if for yourself on Google Trends, you can find a current graph here. The big blue spike centers on the first week of June 2018, just days after the Lightbox announcement and release of the ads.
Colored Diamonds at $800/carat
Element Six, a De Beers-owned company, has been producing synthetic diamond for experimental and industrial purposes since the 1980s. Although De Beers has historically only sold natural diamonds for use in jewelry, in 2018 they surprised the gem and jewelry industry by breaking that tradition. In September 2018 they began selling a collection of synthetic diamond jewelry under the Lightbox trademark. They distinguished their products from natural diamonds by calling then lab-grown diamonds.
Lightbox Jewelry features pink, blue, and colorless lab-grown diamonds for the previously unheard-of price of $800 per carat. These are all sold without grading or laboratory reports. They do not have the lab-grown diamonds graded for two reasons: 1) to minimize the cost of the product; and, 2) they do not believe that lab-grown diamonds require grading. The goal is to sell them inexpensively for any occasion and for everyday wear.
At a price of only $800 per carat, almost anyone who wants a colored lab-grown diamond can afford one. Customers can purchase lab-grown blue or pink diamond solitaire stud earrings starting at $250 per pair. For $250, each earring contains a 1/8-carat lab-grown diamond set in 10-karat white gold.
In May, 2019, Stephen Lussier, a De Beers executive, reported that Lightbox's early sales have been mostly their pink and blue products. The company believes that this is happening because the buyers want "color". They are also selling all of the Lightbox jewelry that they have the ability to produce. [13] In the autumn of 2020, Lightbox opened their new factory in Gresham, Oregon, and it will soon be producing up to 200,000 carats per year. [14]
Mr. Lussier was asked if Lightbox was "cannibalizing the lower end of the diamond market". His reply was "Not significantly. We are selling in a category where it's competing largely with non-diamond jewelry... [We] are actually in a different market segment from a diamond perspective, and there is minimal cannibalization. It will affect some of the semiprecious, the low-end colored stones. But at the end of the market it's mostly color." [13]
| More Diamonds |
 |
Diamond Production |
 |
Red Diamonds |
 |
US Diamond Mines |
 |
Diamonds Do Not Form From Coal |
 |
Diamond |
 |
Colored Diamonds |
 |
Canadian Diamond Mines |
 |
Brown Diamonds |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
