Spodumene
An important source of high-purity lithium and a gemstone with collector appeal
Article by: Hobart M. King, PhD

Color Varieties of Gem-Quality Spodumene: Gem-quality spodumene is named according to its color. Pink to purple specimens are known as kunzite, green specimens are known as hiddenite, and yellow specimens are known as triphane. Clockwise from top left: Purple kunzite from Afghanistan; pink kunzite from Afghanistan; yellowish green hiddenite from the Adams Hiddenite and Emerald Mine in North Carolina; yellow triphane from Afghanistan. Specimens and photos by Arkenstone / www.iRocks.com.
What is Spodumene?
Spodumene is a pyroxene mineral that is typically found in lithium-rich pegmatites. It is usually associated with other lithium minerals such as lepidolite, eucryptite, and petalite. Spodumene has a chemical composition of LiAlSi2O6 but small amounts of sodium sometimes substitute for lithium.
Throughout most of the 20th century, spodumene was the most important ore of lithium metal. Lithium brines discovered in South America and other locations have become a more important source of lithium metal.
Spodumene is also used as a gemstone, and in that use the color variety names of the mineral are used. Pink to purple spodumene is known as kunzite, green spodumene is known as hiddenite, and yellow spodumene is known as triphane.
The perfect cleavage of spodumene makes it a fragile gem for use in rings and any jewelry that might be exposed to abrasion and impact. It is considered a “collector gem” by some gemologists.

Giant Spodumene Crystals: Molds of giant spodumene crystals at the Etta Mines, Black Hills, Pennington County, South Dakota. Note miner at right center for scale. USGS photo. [1]
Enormous Crystals
Spodumene often occurs in extremely large crystals. One of the earliest accounts of large spodumene crystals is from the Etta Mines, Black Hills, Pennington County, South Dakota. The United States Geological Survey, Bulletin 610 reports:
| "The crystals are often of enormous size. In the Etta Mine, where they are best exposed both in the open cut and tunnel, they frequently attain a diameter of 3 to 4 feet and a length of 30 feet. The largest "log" so far found was 42 feet long and 5 feet 4 inches in maximum diameter. This one log alone would yield 90 tons of spodumene." [1] |
Physical Properties of Spodumene |
|
| Chemical Classification | Silicate |
| Color | White, gray, colorless, yellow, green, blue, lilac, pink, brown. Sometimes pleochroic |
| Streak | White, colorless |
| Luster | Vitreous, pearly |
| Diaphaneity | Transparent to translucent |
| Cleavage | Perfect in two directions with parting |
| Mohs Hardness | 6.5 to 7 |
| Specific Gravity | 3.1 to 3.3 |
| Diagnostic Properties | Prismatic crystals with strong striations parallel to their principal axis. Perfect cleavage. |
| Chemical Composition | LiAl(SiO3)2 |
| Crystal System | Monoclinic |
| Uses | Gemstones (kunzite, hiddenite, triphane). Once the most important source of lithium metal. |
Spodumene as an Ore of Lithium
Spodumene once served as the most important ore of lithium metal. It was very costly to liberate lithium from the silicate mineral; however, lithium refined from spodumene was of very high purity. In the late 1900s, subsurface brines with high concentrations of lithium were developed in Argentina, Chile, China and other locations. These brines could be pumped to the surface, allowed to evaporate, and lithium was easily processed from the evaporite material.
As lithium-rich brine deposits were developed, the more costly use of spodumene as an ore of lithium declined. At times, the demand for lithium has exceeded what can be produced from working brine deposits. In those times spodumene can become an important source of lithium metal.

Spodumene: Translucent to transparent spodumene with an attractive pink, yellow, or green color is sometimes faceted, cut en cabochon or used to make tumbled stones. Its perfect cleavage limits its use to jewelry that will not be subject to rough wear or handling. Spodumene is primarily a "collector's gemstone." The larger pieces of spodumene in this image are about one inch long.
Spodumene as a Gemstone
Spodumene sometimes occurs in transparent crystals in pastel shades of pink, purple, green, and yellow. These have been cut into gemstones that are prized by collectors. However, their use in jewelry is limited to pieces that will be subject to limited abuse because of spodumene's perfect cleavage.

Kunzite spodumene: Pink gem-quality spodumene (kunzite) from the Konar Valley, Afghanistan. Creative Commons image by Didier Descouens.
Kunzite
Pink to lilac specimens of gem-quality spodumene are highly prized and known as "kunzite". The color of these specimens is attributed to the presence of manganese as a chromophore. Kunzite is the most commonly encountered spodumene gem.
Many specimens of kunzite are strongly pleochroic, with the deepest color observed when the gem is viewed down the principal axis. To take full advantage of this phenomenon, gemstones are usually cut with their tables perpendicular to the principal axis to yield stones of the deepest color.

Spodumene: A section of an ore-grade spodumene crystal, similar to what might be mined for the production of lithium metal. The specimen is about three inches in length.
Hiddenite
Emerald-green spodumene is known as "Hiddenite." Its vivid green color is very similar to emerald and is attributed to the presence of chromium as a chromophore. It is the rarest gem variety of spodumene. It was first found near the town of White Plains, North Carolina, which changed its name to "Hiddenite" after the popular gemstone that attracted people to the area.

Lithium battery: One of the primary uses of spodumene is in the production of high-purity lithium for use in lithium-ion batteries. The popularity of small electronic devices such as cell phones, portable computers, and cameras is driving the demand for spodumene. Photo copyright iStockphoto / Anton Snarikov.
Triphane
Spodumene rarely occurs in a yellow color. However, some yellow spodumene can be of gem quality, and it has been cut into faceted and cabochon gems. These gems have been given the name “triphane”.
It should be noted that “triphane” is one of the early names used for spodumene. It can be encountered in mineralogical writings from the 1800s and early 1900s. Any use of the word “triphane” from that era is referring to spodumene as a mineral because gemological use of the word did not begin until the late 1900s.
Treatment of Gem-Quality Spodumene
Some gem-quality spodumene will develop a richer color when heated or irradiated. These procedures have been applied to many gems that enter the marketplace. Some of these will fade over time when exposed to direct sunlight. Valuable spodumene gems of any color should be stored away from direct light.
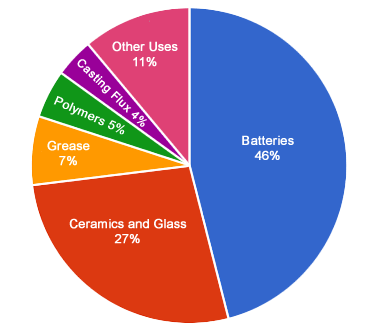
Uses of Lithium: Lithium has many diverse uses. This chart shows estimated global uses of lithium by end product. It is mainly used in manufacturing rechargeable batteries, ceramics, specialty glass, high-temperature grease, polymers, casting flux, aluminum alloys, and pharmaceuticals. USGS data from 2017. [2]
 |
Did You Know? Lithium is an active ingredient in some medications. Salts of lithium are used in medication for bipolar disorder. The lithium contributes to a "mood-stabilizing" effect. One product has been named "Lithium." Image copyright iStockphoto / Paige Foster. |
Demand for Spodumene
The demand for spodumene is dependent upon the use of lithium in manufacturing. In the past, most lithium compounds and minerals were used to produce ceramics, glass, aluminum alloys, and high-temperature grease. However, in the last two decades an exploding demand for rechargeable batteries to power vehicles, cell phones, tablet computers, cameras, music players, GPS units, and other portable electronic devices is driving the demand for high-purity lithium - and that drives the demand for spodumene.
Lithium batteries have a much higher charge-to-weight ratio and power-to-weight ratio than lead/acid and zinc carbon cells. This has increased the demand for lithium as a battery metal. However, the use of batteries to power vehicles has resulted in an enormous amount of research related to battery metals. It is possible that lithium could be replaced by another battery metal such as cobalt.

The best way to learn about minerals is to study with a collection of small specimens that you can handle, examine, and observe their properties. Inexpensive mineral collections are available in the Geology.com Store. Image copyright iStockphoto / Anna Usova.
| Spodumene Information |
|
[1] Mineralogic Notes, Series 3: Waldemar Schaller, Gigantic Crystals of Spodumene, United States Geological Survey, Bulletin 610, 1916. [2] Lithium: Brian W. Jaskula, United States Geological Survey, Mineral Commodity Summaries, January 2018. [3] Lithium: Brian W. Jaskula, United States Geological Survey, Minerals Yearbook, September 2018. |
| More Minerals |
 |
Herkimer Diamonds |
 |
The Acid Test |
 |
Tumbled Stones |
 |
Zircon |
 |
Fool*s Gold |
 |
Kyanite |
 |
Rock Tumblers |
 |
Rhodochrosite |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
