Geology and Resources of Some World Oil-Shale Deposits
Reprint of: USGS Scientific Investigations Report 2005-5294 by John R. Dyni
| What is Oil Shale? |

Oil shale is a rock that contains significant amounts of organic material in the form of kerogen. Up to 1/3 of the rock can be solid organic material. Liquid and gaseous hydrocarbons can be extracted from the oil shale, but the rock must be heated and/or treated with solvents. This is usually much less efficient than drilling rocks that will yield oil or gas directly into a well. The processes used for hydrocarbon extraction also produce emissions and waste products that cause significant environmental concerns.
Oil shale usually meets the definition of "shale" in that it is "a laminated rock consisting of at least 67% clay minerals," however, it sometimes contains enough organic material and carbonate minerals that clay minerals account for less than 67% of the rock.
| United States of America Oil Shale |
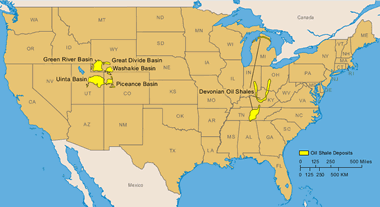
United States: Areas underlain by the Green River Formation in Colorado, Utah, and Wyoming, United States (after Dyni, 2005) and major areas of surface minable Devonian oil shale in the eastern United States (after Matthews and others 1980). More information on United States oil shale. Enlarge map.
| Oil Shale - Country Menu |
Introduction
Oil shale is commonly defined as a fine-grained sedimentary rock containing organic matter that yields substantial amounts of oil and combustible gas upon destructive distillation. Most of the organic matter is insoluble in ordinary organic solvents; therefore, it must be decomposed by heating to release such materials. Underlying most definitions of oil shale is its potential for the economic recovery of energy, including shale oil and combustible gas, as well as a number of byproducts. A deposit of oil shale having economic potential is generally one that is at or near enough to the surface to be developed by open-pit or conventional underground mining or by in-situ methods.
Oil shales range widely in organic content and oil yield. Commercial grades of oil shale, as determined by their yield of shale oil, ranges from about 100 to 200 liters per metric ton (l/t) of rock. The U.S. Geological Survey has used a lower limit of about 40 l/t for classification of Federal oil-shale lands. Others have suggested a limit as low as 25 l/t.
Deposits of oil shale are in many parts of the world. These deposits, which range from Cambrian to Tertiary age, may occur as minor accumulations of little or no economic value or giant deposits that occupy thousands of square kilometers and reach thicknesses of 700 m or more. Oil shales were deposited in a variety of depositional environments, including fresh-water to highly saline lakes, epicontinental marine basins and subtidal shelves, and in limnic and coastal swamps, commonly in association with deposits of coal.
In terms of mineral and elemental content, oil shale differs from coal in several distinct ways. Oil shales typically contain much larger amounts of inert mineral matter (60-90 percent) than coals, which have been defined as containing less than 40 percent mineral matter. The organic matter of oil shale, which is the source of liquid and gaseous hydrocarbons, typically has a higher hydrogen and lower oxygen content than that of lignite and bituminous coal.
In general, the precursors of the organic matter in oil shale and coal also differ. Much of the organic matter in oil shale is of algal origin, but may also include remains of vascular land plants that more commonly compose much of the organic matter in coal. The origin of some of the organic matter in oil shale is obscure because of the lack of recognizable biologic structures that would help identify the precursor organisms. Such materials may be of bacterial origin or the product of bacterial degradation of algae or other organic matter.
The mineral component of some oil shales is composed of carbonates including calcite, dolomite, and siderite, with lesser amounts of aluminosilicates. For other oil shales, the reverse is true-silicates including quartz, feldspar, and clay minerals are dominant and carbonates are a minor component. Many oil-shale deposits contain small, but ubiquitous, amounts of sulfides including pyrite and marcasite, indicating that the sediments probably accumulated in dysaerobic to anoxic waters that prevented the destruction of the organic matter by burrowing organisms and oxidation.
Although shale oil in today's (2004) world market is not competitive with petroleum, natural gas, or coal, it is used in several countries that possess easily exploitable deposits of oil shale but lack other fossil fuel resources. Some oil-shale deposits contain minerals and metals that add byproduct value such as alum [KAl(SO4)2.12H2O], nahcolite (NaHCO3), dawsonite [NaAl(OH)2CO3], sulfur, ammonium sulfate, vanadium, zinc, copper, and uranium.
The gross heating value of oil shales on a dry-weight basis ranges from about 500 to 4,000 kilocalories per kilogram (kcal/kg) of rock. The high-grade kukersite oil shale of Estonia, which fuels several electric power plants, has a heating value of about 2,000 to 2,200 kcal/kg. By comparison, the heating value of lignitic coal ranges from 3,500 to 4,600 kcal/kg on a dry, mineral-free basis (American Society for Testing Materials, 1966).
Tectonic events and volcanism have altered some deposits. Structural deformation may impair the mining of an oil-shale deposit, whereas igneous intrusions may have thermally degraded the organic matter. Thermal alteration of this type may be restricted to a small part of the deposit, or it may be widespread making most of the deposit unfit for recovery of shale oil.
The purpose of this report is to (1) discuss the geology and summarize the resources of selected deposits of oil shale in varied geologic settings from different parts of the world and (2) present new information on selected deposits developed since 1990 (Russell, 1990).
| Australia Oil Shale |
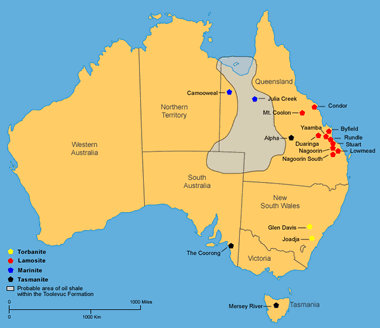
Australia: Deposits of oil shale in Australia (locations after Crisp and others, 1987; and, Cook and Sherwood 1989). More information on Australia oil shale. Enlarge map.
Recoverable Resources
The commercial development of an oil-shale deposit depends upon many factors. The geologic setting and the physical and chemical characteristics of the resource are of primary importance. Roads, railroads, power lines, water, and available labor are among the factors to be considered in determining the viability of an oil-shale operation. Oil-shale lands that could be mined may be preempted by present land usage such as population centers, parks, and wildlife refuges. Development of new in-situ mining and processing technologies may allow an oil-shale operation in previously restricted areas without causing damage to the surface or posing problems of air and water pollution.
The availability and price of petroleum ultimately effect the viability of a large-scale oil-shale industry. Today, few, if any deposits can be economically mined and processed for shale oil in competition with petroleum. Nevertheless, some countries with oil-shale resources, but lack petroleum reserves, find it expedient to operate an oil-shale industry. As supplies of petroleum diminish in future years and costs for petroleum increase, greater use of oil shale for the production of electric power, transportation fuels, petrochemicals, and other industrial products seems likely.
| Brazil Oil Shale |
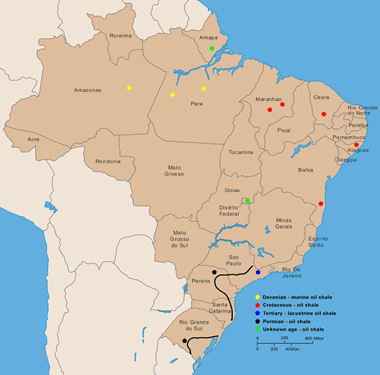
Brazil: Deposits of oil shale in Brazil (locations after Padula, 1969). More information on Brazil oil shale. Enlarge map.
| Canada Oil Shale |
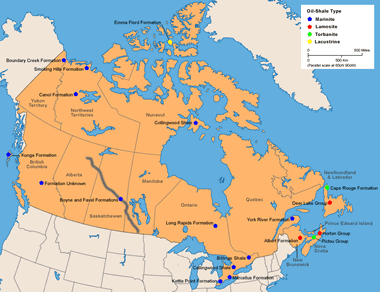
Canada: Oil-shale deposits in Canada (locations after Macauley, 1981). More information on Canada oil shale. Enlarge map.
Determining Grade of Oil Shale
The grade of oil shale has been determined by many different methods with the results expressed in a variety of units. The heating value of the oil shale may be determined using a calorimeter. Values obtained by this method are reported in English or metric units, such as British thermal units (Btu) per pound of oil shale, calories per gram (cal/gm) of rock, kilocalories per kilogram (kcal/kg) of rock, megajoules per kilogram (MJ/kg) of rock, and other units. The heating value is useful for determining the quality of an oil shale that is burned directly in a power plant to produce electricity. Although the heating value of a given oil shale is a useful and fundamental property of the rock, it does not provide information on the amounts of shale oil or combustible gas that would be yielded by retorting (destructive distillation).
The grade of oil shale can be determined by measuring the yield of oil of a shale sample in a laboratory retort. This is perhaps the most common type of analysis that is currently used to evaluate an oil-shale resource. The method commonly used in the United States is called the "modified Fischer assay," first developed in Germany, then adapted by the U.S. Bureau of Mines for analyzing oil shale of the Green River Formation in the western United States (Stanfield and Frost, 1949). The technique was subsequently standardized as the American Society for Testing and Materials Method D-3904-80 (1984). Some laboratories have further modified the Fischer assay method to better evaluate different types of oil shale and different methods of oil-shale processing.
The standardized Fischer assay method consists of heating a 100-gram sample crushed to -8 mesh (2.38-mm mesh) screen in a small aluminum retort to 500ºC at a rate of 12ºC per minute and held at that temperature for 40 minutes. The distilled vapors of oil, gas, and water are passed through a condenser cooled with ice water into a graduated centrifuge tube. The oil and water are then separated by centrifuging. The quantities reported are the weight percentages of shale oil (and its specific gravity), water, shale residue, and "gas plus loss" by difference.
The Fischer assay method does not determine the total available energy in an oil shale. When oil shale is retorted, the organic matter decomposes into oil, gas, and a residuum of carbon char remaining in the retorted shale. The amounts of individual gases-chiefly hydrocarbons, hydrogen, and carbon dioxide-are not normally determined but are reported collectively as "gas plus loss," which is the difference of 100 weight percent minus the sum of the weights of oil, water, and spent shale. Some oil shales may have a greater energy potential than that reported by the Fischer assay method depending on the components of the "gas plus loss."
The Fischer assay method also does not necessarily indicate the maximum amount of oil that can be produced by a given oil shale. Other retorting methods, such as the Tosco II process, are known to yield in excess of 100 percent of the yield reported by Fischer assay. In fact, special methods of retorting, such as the Hytort process, can increase oil yields of some oil shales by as much as three to four times the yield obtained by the Fischer assay method (Schora and others, 1983; Dyni and others, 1990). At best, the Fischer assay method only approximates the energy potential of an oil-shale deposit.
Newer techniques for evaluating oil-shale resources include the Rock-Eval and the "material-balance" Fischer assay methods. Both give more complete information about the grade of oil shale, but are not widely used. The modified Fischer assay, or close variations thereof, is still the major source of information for most deposits.
It would be useful to develop a simple and reliable assay method for determining the energy potential of an oil shale that would include the total heat energy and the amounts of oil, water, combustible gases including hydrogen, and char in sample residue.
| Estonia and Sweden Oil Shale |
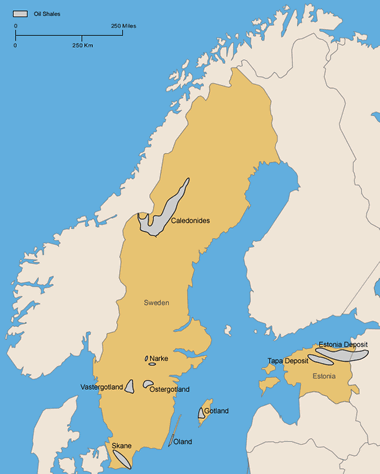
Estonia and Sweden: Location of the kukersite deposits in northern Estonia and Russia (locations after Kattai and Lokk, 1998; and Bauert, 1994). Also, areas of Alum Shale in Sweden (locations after Andersson and others, 1985). More information on Estonia and Sweden oil shale. Enlarge map.
Origin of Organic Matter
Organic matter in oil shale includes the remains of algae, spores, pollen, plant cuticle and corky fragments of herbaceous and woody plants, and other cellular remains of lacustrine, marine, and land plants. These materials are composed chiefly of carbon, hydrogen, oxygen, nitrogen, and sulfur. Some organic matter retains enough biological structures so that specific types can be identified as to genus and even species. In some oil shales, the organic matter is unstructured and is best described as amorphous (bituminite). The origin of this amorphous material is not well known, but it is likely a mixture of degraded algal or bacterial remains. Small amounts of plant resins and waxes also contribute to the organic matter. Fossil shell and bone fragments composed of phosphatic and carbonate minerals, although of organic origin, are excluded from the definition of organic matter used herein and are considered to be part of the mineral matrix of the oil shale.
Most of the organic matter in oil shales is derived from various types of marine and lacustrine algae. It may also include varied admixtures of biologically higher forms of plant debris that depend on the depositional environment and geographic position. Bacterial remains can be volumetrically important in many oil shales, but they are difficult to identify.
Most of the organic matter in oil shale is insoluble in ordinary organic solvents, whereas some is bitumen that is soluble in certain organic solvents. Solid hydrocarbons, including gilsonite, wurtzilite, grahamite, ozokerite, and albertite, are present as veins or pods in some oil shales. These hydrocarbons have somewhat varied chemical and physical characteristics, and several have been mined commercially.
| Israel and Jordan Oil Shale |
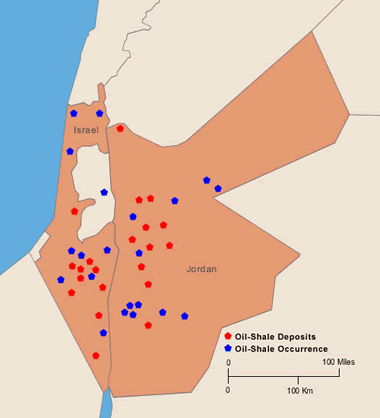
Israel and Jordan: Deposits of oil shale in Israel (locations after Minster, 1994). Also, oil-shale deposits in Jordan (locations after Jaber and others, 1997; and, Hamarneh, 1998). More information on Israel and Jordan oil shale. Enlarge map.
Thermal Maturity of Organic Matter
The thermal maturity of an oil shale refers to the degree to which the organic matter has been altered by geothermal heating. If the oil shale is heated to a high enough temperature, as may be the case if the oil shale were deeply buried, the organic matter may thermally decompose to form oil and gas. Under such circumstances, oil shales can be source rocks for petroleum and natural gas. The Green River oil shale, for example, is presumed to be the source of the oil in the Red Wash field in northeastern Utah. On the other hand, oil-shale deposits that have economic potential for their shale-oil and gas yields are geothermally immature and have not been subjected to excessive heating. Such deposits are generally close enough to the surface to be mined by open-pit, underground mining, or by in-situ methods.
The degree of thermal maturity of an oil shale can be determined in the laboratory by several methods. One technique is to observe the changes in color of the organic matter in samples collected from varied depths in a borehole. Assuming that the organic matter is subjected to geothermal heating as a function of depth, the colors of certain types of organic matter change from lighter to darker colors. These color differences can be noted by a petrographer and measured using photometric techniques.
Geothermal maturity of organic matter in oil shale is also determined by the reflectance of vitrinite (a common constituent of coal derived from vascular land plants), if present in the rock. Vitrinite reflectance is commonly used by petroleum explorationists to determine the degree of geothermal alteration of petroleum source rocks in a sedimentary basin. A scale of vitrinite reflectances has been developed that indicates when the organic matter in a sedimentary rock has reached temperatures high enough to generate oil and gas. However, this method can pose a problem with respect to oil shale, because the reflectance of vitrinite may be depressed by the presence of lipid-rich organic matter.
Vitrinite may be difficult to recognize in oil shale because it resembles other organic material of algal origin and may not have the same reflectance response as vitrinite, thereby leading to erroneous conclusions. For this reason, it may be necessary to measure vitrinite reflectance from laterally equivalent vitrinite-bearing rocks that lack the algal material.
In areas where the rocks have been subjected to complex folding and faulting or have been intruded by igneous rocks, the geothermal maturity of the oil shale should be evaluated for proper determination of the economic potential of the deposit.
| Morocco Oil Shale |
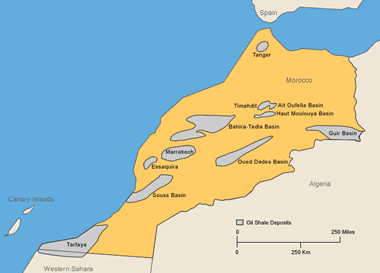
Morocco: Oil-shale deposits in Morocco (locations after Bouchta, 1984). More information on Morocco oil shale. Enlarge map.
Classification of Oil Shale
Oil shale has received many different names over the years, such as cannel coal, boghead coal, alum shale, stellarite, albertite, kerosene shale, bituminite, gas coal, algal coal, wollongite, schistes bitumineux, torbanite, and kukersite. Some of these names are still used for certain types of oil shale. Recently, however, attempts have been made to systematically classify the many different types of oil shale on the basis of the depositional environment of the deposit, the petrographic character of the organic matter, and the precursor organisms from which the organic matter was derived.
A useful classification of oil shales was developed by A.C. Hutton (1987, 1988, 1991), who pioneered the use of blue/ultraviolet fluorescent microscopy in the study of oil-shale deposits of Australia. Adapting petrographic terms from coal terminology, Hutton developed a classification of oil shale based primarily on the origin of the organic matter. His classification has proved to be useful for correlating different kinds of organic matter in oil shale with the chemistry of the hydrocarbons derived from oil shale.
Hutton (1991) visualized oil shale as one of three broad groups of organic-rich sedimentary rocks: (1) humic coal and carbonaceous shale, (2) bitumen-impregnated rock, and (3) oil shale. He then divided oil shale into three groups based upon their environments of deposition - terrestrial, lacustrine, and marine.
Terrestrial oil shales include those composed of lipid-rich organic matter such as resin spores, waxy cuticles, and corky tissue of roots, and stems of vascular terrestrial plants commonly found in coal-forming swamps and bogs. Lacustrine oil shales include lipid-rich organic matter derived from algae that lived in freshwater, brackish, or saline lakes. Marine oil shales are composed of lipid-rich organic matter derived from marine algae, acritarchs (unicellular organisms of questionable origin), and marine dinoflagellates.
Several quantitatively important petrographic components of the organic matter in oil shale-telalginite, lamalginite, and bituminite-are adapted from coal petrography. Telalginite is organic matter derived from large colonial or thick-walled unicellular algae, typified by genera such as Botryococcus. Lamalginite includes thin-walled colonial or unicellular algae that occurs as laminae with little or no recognizable biologic structures. Telalginite and lamalginite fluoresce brightly in shades of yellow under blue/ultraviolet light.
Bituminite, on the other hand, is largely amorphous, lacks recognizable biologic structures, and weakly fluoresces under blue light. It commonly occurs as an organic groundmass with fine-grained mineral matter. The material has not been fully characterized with respect to its composition or origin, but it is commonly an important component of marine oil shales. Coaly materials including vitrinite and inertinite are rare to abundant components of oil shale; both are derived from humic matter of land plants and have moderate and high reflectance, respectively, under the microscope.
Within his three-fold grouping of oil shales (terrestrial, lacustrine, and marine), Hutton (1991) recognized six specific oil-shale types: cannel coal, lamosite, marinite, torbanite, tasmanite, and kukersite. The most abundant and largest deposits are marinites and lamosites.
Cannel coal is brown to black oil shale composed of resins, spores, waxes, and cutinaceous and corky materials derived from terrestrial vascular plants together with varied amounts of vitrinite and inertinite. Cannel coals originate in oxygen-deficient ponds or shallow lakes in peat-forming swamps and bogs (Stach and others, 1975, p. 236-237).
Lamosite is pale- and grayish-brown and dark gray to black oil shale in which the chief organic constituent is lamalginite derived from lacustrine planktonic algae. Other minor components in lamosite include vitrinite, inertinite, telalginite, and bitumen. The Green River oil-shale deposits in western United States and a number of the Tertiary lacustrine deposits in eastern Queensland, Australia, are lamosites.
Marinite is a gray to dark gray to black oil shale of marine origin in which the chief organic components are lamalginite and bituminite derived chiefly from marine phytoplankton. Marinite may also contain small amounts of bitumen, telalginite, and vitrinite. Marinites are deposited typically in epeiric seas such as on broad shallow marine shelves or inland seas where wave action is restricted and currents are minimal. The Devonian-Mississippian oil shales of eastern United States are typical marinites. Such deposits are generally widespread covering hundreds to thousands of square kilometers, but they are relatively thin, often less than about 100 m.
Torbanite, tasmanite, and kukersite are related to specific kinds of algae from which the organic matter was derived; the names are based on local geographic features. Torbanite, named after Torbane Hill in Scotland, is a black oil shale whose organic matter is composed mainly of telalginite derived largely from lipid-rich Botryococcus and related algal forms found in fresh- to brackish-water lakes. It also contains small amounts of vitrinite and inertinite. The deposits are commonly small, but can be extremely high grade. Tasmanite, named from oil-shale deposits in Tasmania, is a brown to black oil shale. The organic matter consists of telalginite derived chiefly from unicellular tasmanitid algae of marine origin and lesser amounts of vitrinite, lamalginite, and inertinite. Kukersite, which takes its name from Kukruse Manor near the town of Kohtla-Järve, Estonia, is a light brown marine oil shale. Its principal organic component is telalginite derived from the green alga, Gloeocapsomorpha prisca. The Estonian oil-shale deposit in northern Estonia along the southern coast of the Gulf of Finland and its eastern extension into Russia, the Leningrad deposit, are kukersites.
| China, Russia, Syria, Thailand, and Turkey |
China, Russia, Syria, Thailand, and Turkey: Other countries with oil shale. More information on China, Russia, Syria, Thailand and Turkey oil shale.
Evaluation of Oil-Shale Resources
Relatively little is known about many of the world's deposits of oil shale and much exploratory drilling and analytical work need to be done. Early attempts to determine the total size of world oil-shale resources were based on few facts, and estimating the grade and quantity of many of these resources were speculative, at best. The situation today has not greatly improved, although much information has been published in the past decade or so, notably for deposits in Australia, Canada, Estonia, Israel, and the United States.
Evaluation of world oil-shale resources is especially difficult because of the wide variety of analytical units that are reported. The grade of a deposit is variously expressed in U.S. or Imperial gallons of shale oil per short ton (gpt) of rock, liters of shale oil per metric ton (l/t) of rock, barrels, short or metric tons of shale oil, kilocalories per kilogram (kcal/kg) of oil shale, or gigajoules (GJ) per unit weight of oil shale. To bring some uniformity into this assessment, oil-shale resources in this report are given in both metric tons of shale oil and in equivalent U.S. barrels of shale oil, and the grade of oil shale, where known, is expressed in liters of shale oil per metric ton (l/t) of rock. If the size of the resource is expressed only in volumetric units (barrels, liters, cubic meters, and so on), the density of the shale oil must be known or estimated to convert these values to metric tons. Most oil shales produce shale oil that ranges in density from about 0.85 to 0.97 by the modified Fischer assay method. In cases where the density of the shale oil is unknown, a value of 0.910 is assumed for estimating resources.
Byproducts may add considerable value to some oil-shale deposits. Uranium, vanadium, zinc, alumina, phosphate, sodium carbonate minerals, ammonium sulfate, and sulfur are some of the potential byproducts. The spent shale after retorting is used to manufacture cement, notably in Germany and China. The heat energy obtained by the combustion of the organic matter in oil shale can be used in the cement-making process. Other products that can be made from oil shale include specialty carbon fibers, adsorbent carbons, carbon black, bricks, construction and decorative blocks, soil additives, fertilizers, rock wool insulating material, and glass. Most of these uses are still small or in experimental stages, but the economic potential is large.
This appraisal of world oil-shale resources is far from complete. Many deposits are not reviewed because data or publications are unavailable. Resource data for deeply buried deposits, such as a large part of the Devonian oil-shale deposits in eastern United States, are omitted, because they are not likely to be developed in the foreseeable future. Thus, the total resource numbers reported herein should be regarded as conservative estimates. This review focuses on the larger deposits of oil shale that are being mined or have the best potential for development because of their size and grade.
| More Rocks |
 |
Tumbled Stones |
 |
Fossils |
 |
Geodes |
 |
The Rock Used to Make Beer |
 |
Topo Maps |
 |
Difficult Rocks |
 |
Fluorescent Minerals |
 |
Sliding Rocks on Racetrack Playa |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
