Talc: The Softest Mineral
What is Talc? How Does it Form? How is Talc Used?
Article by: Hobart M. King, PhD

Uses of talc: Talc is used in a wide variety of products that we see every day. It is an important ingredient in rubber, a filler and whitener in paint, a filler and brightening agent in high-quality papers, and a primary ingredient in many types of cosmetics. Images copyright iStockphoto and (clockwise) MorePixels, Mark Wragg, Franz-W. Franzelin, and High Impact Photography.
Talc: A Mineral in Your Daily Life
Most people are familiar with the mineral known as "talc". It can be crushed into a white powder that is widely known as "talcum powder." This powder has the ability to absorb moisture, absorb oils, absorb odor, serve as a lubricant, and produce an astringent effect with human skin. These properties have made talcum powder an important ingredient in many baby powders, foot powders, first aid powders, and a variety of cosmetics.
A form of talc known as "soapstone" is also widely known. This soft rock is easily carved and has been used to make ornamental and practical objects for thousands of years. It has been used to make sculptures, bowls, countertops, sinks, hearths, pipe bowls, and many other objects.
Although talcum powder and soapstone are two of the more visible uses of talc, they account for a very small fraction of talc consumption. Its hidden uses are far more common. Talc's unique properties make it an important ingredient for making ceramics, paint, paper, roofing materials, plastics, rubber, insecticides, and many other products.

Talc: Talc is a phyllosilicate mineral that cleaves into thin sheets. These sheets are held together only by van der Waals bonds, which allows them to easily slip past one another. This characteristic is responsible for talc's extreme softness, its greasy to soapy feel, and its value as a high-temperature lubricant.
What is Talc?
Talc is a hydrous magnesium silicate mineral with a chemical composition of Mg3Si4O10(OH)2. Although the composition of talc usually stays close to this generalized formula, some substitution occurs. Small amounts of Al or Ti can substitute for Si; small amounts of Fe, Mn, and Al can substitute for Mg; and, very small amounts of Ca can substitute for Mg.
When large amounts of Fe substitute for Mg, the mineral is known as minnesotaite. When large amounts of Al substitute for Mg, the mineral is known as pyrophyllite.
Talc is usually green, white, gray, brown, or colorless. It is a translucent mineral with a pearly luster. It is the softest known mineral and is assigned a hardness of 1 on the Mohs Hardness Scale.
Talc is a monoclinic mineral with a sheet structure similar to the micas. Talc has perfect cleavage that follows planes between the weakly bonded sheets. These sheets are held together only by van der Waals bonds, which allows them to slip past one another easily. This characteristic is responsible for talc's extreme softness, its greasy, soapy feel, and its value as a high-temperature lubricant.
Where is Talc Produced?
| |||||||||||||||||||||||||
In the United States, talc consumption has slowly declined since 1995. Smaller amounts of the mineral are used in the ceramics industry because of a change in firing technology. In the paint industry, a shift from oil-based paints to latex paints has decreased the use of talc. In the cosmetics industry, many manufacturers have replaced talc with cornstarch powder in many products in response to health concerns and litigation. However, the plastics industry is using more talc, as the mineral becomes a more important ingredient in automotive plastics. The amount of talc used in rubber production has also increased slightly.
Mines in the United States have the ability to produce most grades of talc to make the United States self-sufficient. However, some grades of talc can be imported from other countries at lower costs. This reduces employment and revenue in domestic mining and processing companies.
The leading talc-producing countries in 2018 were China, India, Brazil, the United States, South Korea, France, Japan, and Finland. Numerous other countries are minor producers of talc.
The United States, talc is found in the eastern Appalachian and Piedmont regions from New England to Alabama. Important deposits are located in California, Montana, Nevada, Texas and Washington.

Soapstone: A rock known as "soapstone" is a massive variety of talc with varying amounts of other minerals such as micas, chlorite, amphiboles, and pyroxenes. It is a soft rock that is easy to work, which has caused it to be used in a wide variety of dimension stone and sculpture applications. It is used for countertops, electrical panels, hearthstones, figurines, statuary, and many other projects.
How Does Talc Form?
Talc is a mineral that is most often found in the metamorphic rocks of convergent plate boundaries. It forms from at least two processes. Most large talc deposits in the United States formed when heated waters, carrying dissolved magnesium and silica, reacted with dolomitic marbles. A second process of talc formation occurred when heat and chemically active fluids altered rocks such as dunite and serpentinite into talc.
Most of the talc deposits in the United States are in metamorphic rocks on the eastern side of the Appalachian Mountains and in rocks metamorphosed in convergent terranes of Washington, Idaho, Montana, California, Nevada, and New Mexico. Deposits of talc are also found in Texas.

Foliated talc: Talc is a metamorphic mineral that frequently exhibits distinct foliation.
Talc Mining and Processing
Most talc in the United States is produced from an open-pit mine where the rock is drilled, blasted, and partially crushed in the mining operation. The highest grade ores are produced by selective mining and sorting operations.
Great care is taken during the mining process to avoid contaminating the talc with other rock materials. These other materials can have an adverse effect on the color of the product. Contamination can introduce hard particles that cause problems in applications where talc is being used because of its softness or lubricating properties.
Partially crushed rock is taken from the mine to a mill, where it is further reduced in particle size. Impurities are sometimes removed by froth flotation or mechanical processing. The mills usually produce crushed or finely ground talc that meets customer requirements for particle size, brightness, composition, and other properties.
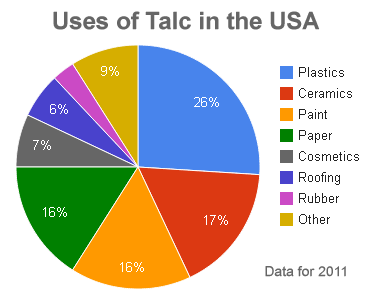
Uses of Talc: Talc is used as a filler, coating, pigment, dusting agent, and extender in plastics, ceramics, paint, paper, cosmetics, roofing, rubber and many other products. Data from the United States Geological Survey.

Talc: Foliated talc that has a black color in massive form but cleaves into thin, flexible, inelastic and colorless sheets.
Physical Properties of Talc |
|
| Chemical Classification | Silicate |
| Color | Green, white, gray, brown, colorless |
| Streak | White to pale green |
| Luster | Pearly |
| Diaphaneity | Translucent |
| Cleavage | Perfect |
| Mohs Hardness | 1 |
| Specific Gravity | 2.7 to 2.8 |
| Diagnostic Properties | Feel, color, softness, cleavage |
| Chemical Composition | Mg3Si4O10(OH)2 |
| Crystal System | Monoclinic |
| Uses | Used as a filler and anti-stick coating in plastics, ceramics, paint, paper, roofing, rubber, cosmetics |

The best way to learn about minerals is to study with a collection of small specimens that you can handle, examine, and observe their properties. Inexpensive mineral collections are available in the Geology.com Store. Image copyright iStockphoto / Anna Usova.
| Talc Information |
|
[1] Talc and Pyrophyllite: Robert L. Virta, U.S. Geological Survey, Mineral Commodity Summaries, January 2012.
[2] Talc and Pyrophyllite: Robert L. Virta, U.S. Geological Survey, 2010 Minerals Yearbook, October 2012. [3] U.S. Talc -- Baby Powder and Much More: U.S. Geological Survey, Fact Sheet FS-065-00, September 2000. [4] Talc resources of the conterminous United States: Robert C. Greene, United States Geological Survey, Open-File Report OF 95-586, 1995. [5] Talc in Cosmetics: United States Food and Drug Administration, website article, last accessed August 2022. |
Uses of Talc
Most people use products made from talc every day; however, they don't realize that talc is in the product - or the special role that it plays.
Talc in Plastics
In 2011, about 26% of the talc consumed in the United States was used in the manufacturing of plastics. It is mainly used as a filler. The platy shape of talc particles can increase the stiffness of products such as polypropylene, vinyl, polyethylene, nylon, and polyester. It can also increase the heat resistance of these products and reduce shrinkage. Where the plastic is extruded in the manufacturing process, talc's very low hardness produces less abrasion on equipment than harder mineral fillers.
Talc in Ceramics
In the United States in 2011, about 17% of the talc consumed was used in the manufacturing of ceramics products such as bathroom fixtures, ceramic tile, pottery, and dinnerware. When used as a filler in ceramics, talc can improve the firing characteristics of the greenware and the strength of the finished product.
Talc in Paint
Most paints are suspensions of mineral particles in a liquid. The liquid portion of the paint facilitates application, but after the liquid evaporates, the mineral particles remain on the wall. Talc is used as an extender and filler in paints. The platy shape of talc particles improves the suspension of solids in the can and helps the liquid paint adhere to a wall without sagging.
Powdered talc is a very bright white color. This makes talc an excellent filler in paint because it simultaneously serves to whiten and brighten the paint. Talc's low hardness is valued because it causes less abrasion damage on spray nozzles and other equipment when paint is applied. In 2011, about 16% of the talc consumed in the United States was used to make paint.
Talc in Paper
Most papers are made from a pulp of organic fibers. This pulp is made from wood, rags, and other organic materials. Finely ground mineral matter is added to the pulp to serve as a filler. When the pulp is rolled into thin sheets, the mineral matter fills spaces between the pulp fibers, resulting in a paper with a much smoother writing surface. Talc as a mineral filler can improve the opacity, brightness, and whiteness of the paper. Talc can also improve the paper's ability to absorb ink. In 2011, the paper industry consumed about 16% of the talc used in the United States.
Talc in Cosmetics and Antiperspirants
Finely ground talc is used as the powder base of many cosmetic products. The tiny platelets of a talc powder readily adhere to the skin but can be washed off easily. Talc's softness allows it to be applied and removed without causing skin abrasion.
Talc also has the ability to absorb oils and perspiration produced by human skin. The ability of talc to absorb moisture, absorb odor, adhere to the skin, serve as a lubricant, and produce an astringent effect in contact with human skin make it an important ingredient in many antiperspirants. In 2011, about 7% of the talc consumed in the United States was used to make cosmetics and antiperspirant.
Talc and asbestos occur naturally and may occur in close proximity in some metamorphic rocks. Studies published in the 1960s and 1970s identified health concerns about the use of talc that contains asbestos in some cosmetic products.
According to the FDA, "These studies have not conclusively demonstrated such a link, or if such a link existed, what risk factors might be involved." To address these concerns, talc mining sites are now carefully selected, and ores are carefully processed to avoid the presence of asbestos in talc destined for use in the cosmetics industry.
Talc in Roofing Materials
Talc is added to the asphaltic materials used to make roofing materials to improve their weather resistance. It is also dusted onto the surface of roll roofing and shingles to prevent sticking. In 2011, about 6% of the talc consumed in the United States was used to manufacture roofing materials.
Dimension Stone
A rock known as "soapstone" is a massive variety of talc with varying amounts of other minerals such as micas, chlorite, amphiboles, and pyroxenes. It is a soft rock that is easy to work, which has caused it to be used in a wide variety of dimension stone and sculpture applications. It is used for countertops, electrical panels, hearthstones, figurines, statuary, and many other projects.
Other Uses of Talc
Ground talc is used as a lubricant in applications where high temperatures are involved. It is able to survive at temperatures where oil-based lubricants would be destroyed.
Talc powder is used as a carrier for insecticides and fungicides. It can easily be blown through a nozzle and readily sticks to the leaves and stems of plants. Its softness reduces wear on application equipment.
| More Minerals |
 |
Herkimer Diamonds |
 |
The Acid Test |
 |
Tumbled Stones |
 |
Zircon |
 |
Fool*s Gold |
 |
Kyanite |
 |
Rock Tumblers |
 |
Rhodochrosite |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
