Home » Rocks » Sedimentary Rocks » Rock Salt
Rock Salt
A sedimentary rock composed of the mineral halite.
Article by: Hobart M. King, PhD

Rock Salt: A piece of rock salt composed of many individual salt crystals. The gray color in parts of the specimen is caused by mineral or hydrocarbon impurities. The specimen is estimated to be about three inches across. Photo by Shawn Hempel / Alamy Stock Photo.
What is Rock Salt?
Rock salt is the name of a sedimentary rock that consists almost entirely of halite, a mineral composed of sodium chloride, NaCl. It forms where large volumes of sea water or salty lake water evaporate from an arid-climate basin -- where there is a replenishing flow of salt water and a restricted input of other water.
Deposits of rock salt occur as laterally extensive rock units in many parts of the world. These water-soluble rocks are rarely found at Earth's surface, except in arid countries. The most geographically extensive salt layers in the United States underlie thousands of square miles and reach thicknesses of over 100 feet.
People have mined rock salt or produced salt by evaporation for thousands of years. Most of the ancient salt production was used in cooking and food preservation.
Today in the United States, highway deicing is the most important use of rock salt. Rock salt is also used in chemical manufacturing, food processing, agriculture, plastics, paint, textiles, leather processing, water treatment, and many other industries.
Table of Contents
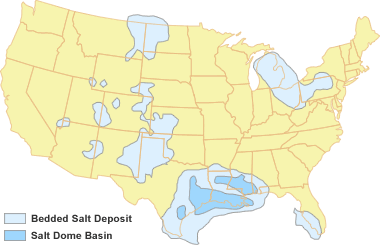
U.S. Salt Deposits: This map shows the location of bedded salt deposits in the United States. The large continuous deposit along the Gulf Coast contains three basins where salt domes have developed. Map by Geology.com with locational data from: Salt Caverns and Their Use for Disposal of Oil Field Wastes, a brochure produced by The National Petroleum Technology Office, Argonne National Laboratory, 1999.
How Does Rock Salt Form?
Deposits of rock salt thick enough for underground mining or solution mining form under a rare set of geological conditions. The deposits shown on the accompanying map formed during times of high sea level, when shallow seas spread over extensive areas of continental crust. To deposit a thick layer of salt, long periods of sea water evaporation had to occur in an arid climate, in basins where there was a continuous or repeated influx of new ocean water.
Over 100 feet of ocean water must be evaporated to produce a single foot of salt. So, to produce a salt layer that is 50 feet thick, over one mile of sea water would have been evaporated. The water was not a mile deep. Instead, there was a continuous - or a repeated - inflow of salt water from the ocean into the salt-forming basin - which was in an arid area and subsiding at a rate that allowed the accumulation of a thick salt.
The formation of thick salt deposits in these basins was a marvel of nature.

Color in Rock Salt: A specimen of rock salt from Bischofferode, Thuringia, Germany. Red, pink, or brown color in salt can be caused by iron oxide stains or trace elements. Blue color sometimes results from defects in the salt's crystal structure known as color centers. Photo by Björn Wylezich / Alamy Stock Photo.

Rock Salt at a Spice Market: Rock salt of various colors offered for sale at a spice shop in the Chandni Chowk markets in Old Delhi, India. Photo by Christopher Bellette / Alamy Stock Photo.
What Color Is Rock Salt?
Pure rock salt under bright illumination will range in color between colorless and white. Colorless salt is usually the most pure because the most common cause of color is impurities. White salt often contains minute gas-filled or fluid-filled cavities.
Specimens or zones of other colors can be caused by mineral grains included in the salt, trace elements in the salt, or small defects in the salt's crystal structure - such as a missing electron. The remains of microscopic organisms in the salt can also produce color.
Most specimens of raw rock salt contain minute to clearly visible inclusions. These can be clastic sediment particles, such as clay minerals; crystals of secondary minerals, such as sylvite; or, patches of solid or liquid hydrocarbons. Crystals or massive inclusions of other evaporite minerals, such as gypsum, anhydrite, or sylvite are common.
Reflection of light by any included material can impart an apparent color to the salt. Absorption of light by included materials can produce an apparent gray or black color. Trace elements or defects in the salt's crystal structure, known as color centers, sometimes produce color in the salt.

Specialty Salts: A chef might want to use specialty salts because of their different colors, textures, and tastes. The salt sampler shown contains rose salt from Bolivia, reddish brown Alpine salt from Austria, pink Himalayan salt from Pakistan, Persian blue salt from Iran, clear halite from Pakistan, and black kala namak salt from India. Photo by Eleventh Hour Photography / Alamy Stock Photo.
Specialty Salts in Cooking
If you visit a store where cooking supplies and spices are sold, you might see salt in a wide variety of colors and textures being sold. Many of these "specialty salts" are natural materials. Others have been crystallized by people or processed to make a distinctive product.
Salt is sometimes crystallized to produce flake-shaped grains which have a unique texture or intensity of flavor. Some are ground coarsely to produce a burst of saltiness as they are crushed between your teeth. Some are ground finely so that they dissolve quickly, season uniformly, or coat the parts of a food particle that is most likely to touch your tongue. There is more to salt than one might imagine.
Some salts are enhanced with spices, flavoring, or color to produce a special dining experience. If you are purchasing a packaged product, check the description and ingredients to learn fascinating facts about the salt.
Some black cooking and table salts contain small amounts of activated charcoal which produce a flavor that many people enjoy. Pink salts are the most popular. Their color is often natural and caused by trace amounts of iron or another element included in the salt. Some have been artificially colored by people.
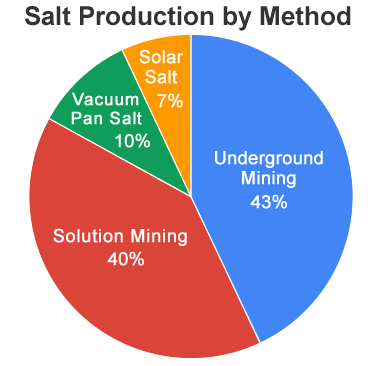
Salt Production Methods: The pie diagram above shows the approximate amount of rock salt (and rock salt equivalent) produced in the United States during the calendar year 2020. Underground mining and solution mining together accounted for about 83% of the salt. Data from the United States Geological Survey Mineral Commodity Summary for 2021.
Rock Salt Production
In 2020 about 39 million tons of salt were produced in the United States. There are four important categories of rock salt production:
- Underground mining
- Solution mining
- Vacuum pan salt
- Solar Salt
The United States consumes more salt than it produces. To satisfy demand in 2020, about 16 million tons of salt were imported.
The amount of imported salt has been increasing in the past few decades. This is mostly a result of increasing demand in the United States and lower production costs in other countries.

Salt Core: Photograph of a short segment of a salt core, obtained by drilling a well down to a subsurface salt layer and retrieving a cylinder of the salt. A core of the entire salt layer is often obtained and brought to the surface for examination by a geologist, and for chemical and physical testing. The properties of numerous salt cores will be used to determine which portion of the rock layer will be mined. Photo by MXWB Photography / Alamy Stock Photo.
Underground Mining
Companies interested in developing a salt resource located hundreds to thousands of feet below the surface usually drill numerous wells down to and through the salt layer. They drill to learn the thickness of the salt and what types of rocks enclose it.
They also obtain core samples of the salt (see accompanying photo) that will be used to determine its chemical and mineral composition. The purity of the salt determines how it can be used. The depth determines the cost to build the mine. Depth also determines the electricity costs of operation, ventilation, and lifting salt, equipment, people, and water in and out of the mine.
Most of the rock salt produced in the United States is produced by traditional room and pillar mining - a mining method that is widely employed in mining for coal. This involves sending people and machines underground to remove the salt. The salt is usually hundreds of feet below Earth's surface.
To start the mine, a large-diameter shaft is drilled vertically down to the salt layer. That shaft will be equipped with lifts, much like elevators, that will be used to lower equipment, people, and supplies down to the level of the salt. Other shafts will be built down to the salt. Some of these shafts will be used to lift the mined salt up to the surface. Others will be used to bring fresh air into the mine or exhaust mine air to the surface.

Continuous Mining Machine: The machine in the photo above is a continuous miner in a salt mine. The cutting head on the right side of the photo is a rotating drum equipped with durable cutting points. As the drum rotates, the cutting head grinds the salt into small pieces which fall onto a pan directly below. Photograph taken at the Salt Union Mine, Winsford, Cheshire, UK. Photo by Jason Bye / Alamy Stock Photo.
Once they have been lowered down a shaft to the level of the salt, the mining machines will begin cutting underground tunnels through the salt. A large cylindrical drum with cutting picks is mounted on the front of the mining machine. This drum rotates and cuts its way through the salt (see accompanying photo).
As a mining machine cuts its way through the salt, broken pieces of salt fall onto a large metal pan mounted immediately below the rotating cutting drum. Mechanized arms rake the salt up the pan and onto a conveyor that carries the salt to a waiting wagon or to a mobile conveyor system. The conveyor will transport the salt to a mine shaft, where the salt will be lifted to the surface, or to an underground storage area. This is the basic process of underground salt mining with a continuous mining machine.
Solution Mining
Solution mining of salt is done by injecting hot water under pressure down a well into a subsurface layer of rock salt. That same water is then withdrawn up to the surface through a nearby recovery well. While the water travels through the layer of rock salt - from the injection well to the recovery well - it dissolves a significant amount of salt. The water is returned to the surface as a concentrated salt solution known as "brine".
Most solution mining sites in the United States are operated by chemical companies who use the brine as a feedstock at a chemical manufacturing plant a short distance from the recovery well. A few solution mining sites have ponds at the surface where they produce solar salt. Others use heat or a vacuum heating process to recrystallize the salt. These recrystallized salts have a higher purity than salt mined from the same rock unit. The recrystallized salt is a higher quality and higher purity because insoluble mineral matter and other insoluble impurities were left in the ground.

Vacuum Pan Crystallizers: Six vacuum pan crystallizers connected in a series. They are approximately 30 feet in height. These crystallizers are in a sugar plant, but similar units are used in salt crystallization. Photo by Westend61 GmbH / Alamy Stock Photo.
Vacuum Pan Salt
Vacuum pan salt is produced in large enclosed tanks known as vacuum pans or vacuum salt crystallizers. The tanks are filled with brine, which is heated by injecting steam into the tank. The steam heats the brine and causes it to boil. As the brine boils it produces additional steam, which is fed into a second vacuum pan and causes its water to boil. Three or more vacuum pans are connected in a series to make efficient use of the steam. As the brine in the tanks becomes saturated with salt, crystals of salt begin to form in the tanks.
Salt produced in a vacuum pan has a much higher purity than salt produced from an underground mine. Why? Vacuum pan salt is usually made from brine produced by solution mining. When the salt was dissolved underground, solid particle impurities in the salt were not dissolved and remained underground.
Vacuum pan salt also has a different texture - its crystals are very small. Some vacuum pan salt also has a different crystal shape - flake-shaped crystals instead of cubic-shaped crystals. Flake salt is a specialty product sold for table use. It is also preferred by many producers of baked goods, butter, cheese, and other products (Date from the United States Geological Survey, Minerals Yearbook for 2016.

Artisanal Solar Salt Production: Workers at a salt pan near Laem Pak Bia, Thailand, harvesting solar salt by manual labor. Photo by Michael McKee / Alamy Stock Photo.

Mechanized Solar Salt Production: Heavy equipment is used to construct salt pans and to harvest their thick layers of salt. This bulldozer is used on a solar salt plantation on Bonaire, an island of the Leeward Antilles, in the Caribbean Sea. Photo by Roland & Renate Kraft / Alamy Stock Photo.
Solar Salt
People have been producing solar salt from ocean water for thousands of years. Solar salt can be produced in parts of the world where evaporation rates significantly exceed precipitation rates. The name "solar" means that the sun acts as a heat source to evaporate ocean water (or brine brought up from the subsurface), leaving behind its dissolved salt.
Historically, the salt was produced in shallow ponds, built along the seashore with inlets just below the level of high tide. Then, twice each day, at the instant that high tide reaches its peak, water flows into the ponds to fill them with salt-laden water. Then for the next 12 hours, the water evaporates to produce salt. This process has been operated as an industry for thousands of years.
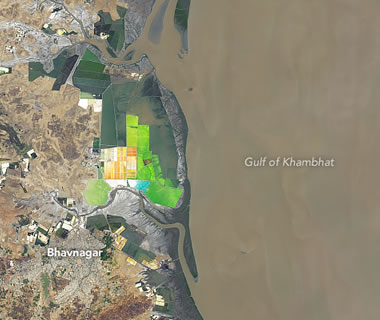
Indian Salt Production: Nearly 75% of India's salt is produced by hand in the state of Gujarat. The colorful rectangles are evaporation pans. Image by Lauren Dauphin, NASA Earth Observatory.
How much salt can be made? That depends upon the climate, the weather, the surface area of the ponds, how they are built - and the knowledge of the people who tend them. For each liter of water they evaporate, about 35 grams of salt (about six teaspoons) can potentially be recovered.
In the United States a few million tons of solar salt is produced each year. Arizona, California, Nevada, New Mexico, Oklahoma, and Utah are important solar salt producers. Brazil, Mexico, and India produce more solar salt than the United States.
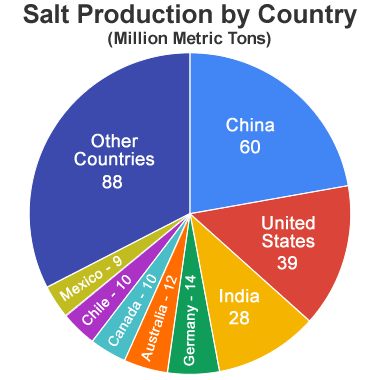
Salt-Producing Countries: China, the United States, and India are the leading producers of rock salt. However, most other countries have domestic salt production. In India, production of salt by artisanal methods is an important source of employment even though the work pays little, is extremely demanding, and exposure to sun and salt water takes a heavy toll on the workers. Estimated salt production by country from the United States Geological Survey Mineral Commodity Summary for 2021.
Rock Salt Producers
The United States Geological Survey Mineral Commodity Summaries for 2021 reports that "almost every country in the world has salt deposits or solar evaporation operations of various sizes". [1]
Eight countries (China, the United States, India, Germany, Australia, Canada, Chile, and Mexico) each produced at least 9 million tons of salt in calendar year 2020. The accompanying pie chart illustrates their relative importance. Much of the salt produced in India is solar salt made using artisanal methods.
Unlike most other mineral commodities, the natural salt resource is enormous. Ocean water can provide an essentially unlimited amount of salt. The amount of salt held in subsurface rock units is enough to easily supply the world for centuries.
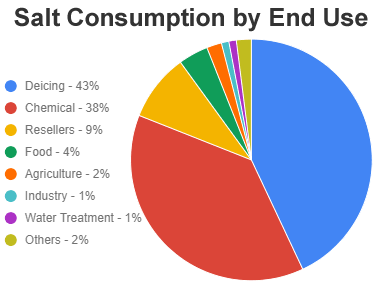
Salt Consumption in the United States: The chart above shows the general categories of salt consumption in the United States. Highway deicing and chemical manufacturing are the most important uses of salt.
Uses of Rock Salt
Rock salt has many uses. The most important uses in the United States are described below.

Highway Deicing: A truck spreading rock salt on an asphalt highway in the United Kingdom. Rock salt and brine are applied to highways in many countries where ice and snow can create a hazard. Photo by Lubo Ivanko / Alamy Stock Photo.
Highway Deicing
The leading use of rock salt in the United States is highway deicing. In calendar year 2020, an estimated 43% of rock salt consumption was used for this purpose. The amount of highway salt consumed varies significantly from year to year, depending upon weather conditions. Variations in the need for highway salt are responsible for most of the year-to-year variation in national consumption.
The rock salt is mined, crushed to appropriate size, and usually applied to roads and highways without further processing. When applied dry, much of the salt can bounce off of the road or be scattered by the wind of passing traffic. If the salt is applied wet, more of it sticks to the road, and the loss of salt is reduced.
Use of rock salt as a road and highway deicer has some environmental problems. Two are related to salt dissolved in water entering the ground on the roadside. The salty water can kill roadside vegetation. It can also contaminate shallow groundwater that might be withdrawn from personal or private water supply wells. It might also discharge into local streams where it can kill or sicken plants, fish, and animals.

Soda Ash: Salt is one of the raw materials used to make soda ash at this chemical plant in Sowa, Botswana. Photo by Karin Duthie / Alamy Stock Photo.
Chemical Industry Feedstock
The second most important use of salt in the United States is as a feedstock for the chemical industry. In 2020, about 38% of the country's salt consumption was used to produce chlorine, sodium hydroxide, synthetic soda ash, and other chemicals. The chemicals were then used as ingredients in plastics, paints, solvents, and a variety of household and industrial cleaning products.
Instead of producing their salt by traditional mining methods, the chemical industry obtains much of their salt by solution mining. They drill wells down into a subsurface salt layer and pump water down some of the wells. That water dissolves some of the salt and is withdrawn up other wells as a concentrated salt solution.
That salt solution, known as brine, is rich in sodium and chlorine and is used in chemical manufacturing. The map at the top of this page shows the geographic extent of the most important parts of the United States' subsurface salt resource.

Sidewalk Salt: A National Park Service worker spreads salt on sidewalks at the White House. Photo by Jonathan Ernst / Reuters / Alamy Stock Photo.
Retail and Wholesale Sales
Bags of rock salt are sold by wholesale and retail stores in many countries where the climate can cause snow and ice problems on roads, sidewalks, driveways, and parking areas. Homeowners and businesses buy much of this salt.
In 2020 about 9% of the rock salt consumed in the United States was sold in retail and wholesale transactions. Much was also sold in bulk and used for pavement deicing by local snow removal companies and local governments. This salt was also sold for a diversity of other uses.

Gourmet Salts: Most people use standard table salt on their food and in their cooking. However, if you visit a gourmet food store, you might be amazed at the variety of salts that are sold. Salts with different grain sizes, grain shapes, additives and origins can have very different tastes. Coarse salts shown in the photo are intended for cooking; however, they can be ground to a smaller particle size for table use or placed in a shaker with a built-in grinder that allows diners to grind the salt at their table. Photo by Andrii Gorulko / Alamy Stock Photo.
Food Processing
Almost every person in the United States consumes some salt in every meal. Even if they do not use a salt shaker - the salt was probably added when the food was processed. In 2020, about 4% of the salt consumed in the United States was used by the food processing industry and seasoning makers.
Salt is used in food to enhance flavor, to enhance color, as a preservative, to enhance texture, to control fermentation, to tenderize, and for other reasons. Pets need salt as an essential nutrient, and a significant amount of salt is used to make pet foods.

Salt Block: Animals licking a salt block is a common sight on farms in the United States. Salt blocks are used to supplement the mineral content of the animal's grazing and feed diet. Photo by Sari O'Neal / Alamy Stock Photo.
Agricultural Salt
Salt is an essential nutrient of farm animals. In 2020 about 2% of the United States' salt consumption was sold to the agricultural industry. Salt is used in preparing some animal feeds, as a water conditioner, and as salt blocks.
Salt blocks are 50-pound cubes of salt that are placed in animal feeding or grazing areas. The animals instinctively lick them to satisfy their need for salt. Many salt blocks have added iodine, sulfur, trace elements, and vitamins to make up for nutrients that might not be received by the animal in their grazing and feed.
| Rock Salt Information |
|
[1] Salt: by Wallace P. Bolen, United States Geological Survey, Mineral Commodity Summaries, January 2021.
|
Water Treatment
Salt is used in commercial and residential water-conditioning units to treat hard water - a problem that occurs when water contains an excessive amount of calcium and magnesium ions. Hard water can cause a build-up of mineral deposits in pipes, equipment, and appliances. It can also reduce the effectiveness of soap and other cleaning supplies.
Water conditioning systems expose the water to sodium ions obtained from the salt. The sodium ions are exchanged for the calcium and magnesium ions in the water to reduce hardness. The salt supply in these water conditioning systems must periodically be replenished.
| More Rocks |
 |
Tumbled Stones |
 |
Fossils |
 |
Geodes |
 |
The Rock Used to Make Beer |
 |
Topo Maps |
 |
Difficult Rocks |
 |
Fluorescent Minerals |
 |
Sliding Rocks on Racetrack Playa |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|

