Home » Minerals » What is a Mineral?
What are Minerals?
Minerals are the foundation of the construction, manufacturing and agricultural industries.
Article by: Hobart M. King, PhD

Mineral Crystals: The beautiful purple crystals in the photo above are examples of amethyst, a purple color variety of quartz. These amethyst crystals grew in a cavity below the earth and because they grew in an open space they were able to form into six-sided prisms with pointed terminations. The prismatic habit is characteristic of amethyst. Image copyright iStockphoto / MvH.
We Use Minerals Every Day!
Every person uses products made from minerals every day. The salt that we add to our food is the mineral halite. Antacid tablets are made from the mineral calcite. Rubies and sapphires are colored varieties of a mineral named corundum.
It takes many minerals to make something as simple as a wooden pencil. The "lead" is made from graphite and clay minerals, the brass band is made of copper and zinc, and the paint that colors it contains pigments and fillers made from a variety of minerals. A cell phone is made using dozens of different minerals that are sourced from mines throughout the world.
The cars that we drive, the roads that we travel, the buildings that we live in, and the fertilizers used to produce our food are all made using minerals.
In the United States, about three billion tons of mineral commodities are consumed each year to support the standard of living of 300 million citizens. That is about ten tons of mineral materials consumed for every person, every year.

Common items made from minerals: Most of the things that we use in our daily life are either made from minerals or produced using mineral products. Antacid tablets are made from calcite, table salt is crushed halite, several minerals are used to make a wood pencil, and dozens of minerals from many different countries are used to make a cell phone.
 |
Did You Know? The white "m" on a piece of M&M's candy is a titanium oxide pigment, most likely produced from the mineral rutile. |
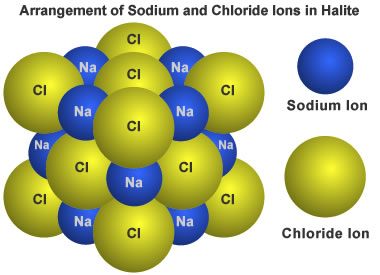
Structure of the mineral halite: The mineral "halite" has a chemical composition of NaCl. That means it contains equal numbers of sodium and chloride atoms. In this case they are electrically charged atoms, known as ions. Those ions are arranged in a cubic pattern that repeats in all directions. The small sodium ions are positioned between the larger chloride ions.
What are Minerals?
To meet the definition of "mineral" used by most geologists, a substance must meet five requirements:
 naturally occurring naturally occurring inorganic inorganic solid solid definite chemical composition definite chemical composition ordered internal structure ordered internal structure |
"Naturally occurring" means that people did not make it. Steel is not a mineral because it is an alloy produced by people.
"Inorganic" means that the substance is not made by an organism. Wood and pearls are made by organisms and thus are not minerals.
"Solid" means that it is not a liquid or a gas at standard temperature and pressure. Water is not a mineral because it is a liquid.
"Definite chemical composition" means that all occurrences of that mineral have a chemical composition that varies within a specific limited range. For example: the mineral halite (known as "rock salt" when it is mined) has a chemical composition of NaCl. It is made up of an equal number of atoms of sodium and chlorine.
"Ordered internal structure" means that the atoms in a mineral are arranged in a systematic and repeating pattern. The structure of the mineral halite is shown in the illustration on this page. Halite is composed of an equal ratio of sodium and chlorine atoms arranged in a cubic pattern.
 |
Did You Know? Although liquid water is not a mineral, it is a mineral when it freezes. Ice is a naturally occurring, inorganic solid with a definite chemical composition and an ordered internal structure. Learn more. |
The Word "Mineral"
The word "mineral" is used in many different ways. Here are some examples:
Geologist's Definition
A formal definition of a mineral, as used by geologists would be: A naturally occurring inorganic solid that has a definite chemical composition, and an ordered internal structure. Geologists are able to identify minerals because they have characteristic physical properties.
Nutritionist's Definition
The word "mineral" also has a nutritional meaning, which is different from the meaning used by geologists.
A nutritionist uses the word mineral when referring to the many inorganic substances that organisms need to grow, repair tissue, metabolize, and carry out other body processes. Mineral nutrients for the human body include: iron, calcium, copper, sulfur, phosphorus, magnesium and many others.

Rhodochrosite: Specimen of rhodochrosite from the Sunnyside Mine, San Juan County, Colorado. Rhodochrosite is a manganese carbonate mineral (MnCO3) that is used as an ore of manganese and is also cut as a gemstone. USGS image.
Archaic Use of "Mineral"
An archaic use of the word "mineral" comes from the Linnaean taxonomy in which all things can be assigned to the animal, vegetable, and mineral kingdoms.

Minerals in rocks: Most rocks are aggregates of minerals. This rock, a granite pegmatite, is a mixture of mineral grains. It contains pink orthoclase, milky quartz, black hornblende and black biotite.
Inconsistent Use of "Mineral"
The word "mineral" is also used inconsistently. In mining, anything obtained from the ground and used by man is considered to be a "mineral commodity" or a "mineral material." These include: crushed stone, which is a manufactured product made from crushed rocks; lime, which is a manufactured product made from limestone or marble (both composed of the mineral calcite); coal which is organic; oil and gas which are organic fluids; rocks such as granite that are mixtures of minerals; and, rocks such as obsidian which are mineraloids and do not have a definite composition and ordered internal structure.
| 2019 United States Mineral Commodity Consumption | ||
| Mineral Commodity | Million Metric Tons | |
| Crushed Stone | 1,600 | |
| Sand and Gravel | 980 | |
| Cement | 102 | |
| Salt | 57.0 | |
| Gypsum | 42.0 | |
| Iron Ore | 41.0 | |
| Phosphate Rock | 25.0 | |
| Clays | 22.0 | |
| Lime | 18.0 | |
| Sulfur | 9.4 | |
| Potash | 5.4 | |
| Soda Ash | 5.2 | |
| Barite | 3.0 | |
| Copper | 1.8 | |
| Lead | 1.6 | |
| ||
Mineral Commodities in Industry
The construction industry is the largest consumer of mineral commodities. Crushed stone is used for foundations, road base, concrete, and drainage. Sand and gravel are used in concrete and foundations. Clays are used to make cement, bricks, and tile. Iron ore is used to make reinforcing rods, steel beams, nails, and wire. Gypsum is used to make drywall. Dimension stone is used for facing, curbing, flooring, stair treads, and other architectural work. These are just a few of the many uses for these commodities in construction.
In agriculture, phosphate rock and potash are used to make fertilizer. Lime is used as an acid-neutralizing soil treatment. Mineral nutrients are added to animal feed.
The chemical industry uses large amounts of salt, lime, and soda ash. Large amounts of metals, clay, and mineral fillers/extenders are used in manufacturing.
 |
Mohs Hardness Scale is a set of reference minerals used for classroom hardness testing. Determining the hardness of a mineral is one of the most important tests used in mineral identification. |

The best way to learn about minerals is to study with a collection of small specimens that you can handle, examine, and observe their properties. Inexpensive mineral collections are available in the Geology.com Store. Image copyright iStockphoto / Anna Usova.
 |
The Acid Test: Geologists use dilute hydrochloric acid to identify carbonate minerals. Carbonate minerals will effervesce with various levels of vigor in contact with the acid. |
Physical Properties of Minerals
There are approximately 4000 different minerals, and each of those minerals has a unique set of physical properties. These include: color, streak, hardness, luster, diaphaneity, specific gravity, cleavage, fracture, magnetism, solubility, and many more. These physical properties are useful for identifying minerals. However, they are much more important in determining the potential industrial uses of the mineral. Let's consider a few examples.
The mineral talc, when ground into a powder, is perfectly suited for use as a foot powder. It is a soft, slippery powder so it will not cause abrasion. It has the ability to absorb moisture, oils, and odor. It adheres to the skin and produces an astringent effect - yet it washes off easily. No other mineral has a set of physical properties that are as suitable for this purpose.
The mineral halite, when crushed into small grains, is perfectly suited for flavoring food. It has a salty taste that most people find pleasing. It dissolves quickly and easily, allowing its flavor to spread through the food. It is soft, so if some does not dissolve it will not damage your teeth. No other mineral has physical properties that are better suited for this use.
The mineral gold is perfectly suited for use in jewelry. It can be easily shaped into a custom item of jewelry by a craftsperson. It has a pleasing yellow color that most people enjoy. It has a bright luster that does not tarnish. Its high specific gravity gives it a nice "heft" that is preferred by most people over lighter metals. Other metals can be used to make jewelry, but these properties make gold an overwhelming favorite. (Some people might add that gold's rarity and value are two additional properties that make it desirable for jewelry. However, rarity is not a property, and its value is determined by supply and demand.)

Star sapphire: A deep blue star sapphire 8 mm x 6 mm cabochon from Thailand. Inclusions of rutile within the stone align with the crystallographic axis of the corundum to produce the star - which is only clearly visible and centered when the back of the stone is cut at 90 degrees to the C-axis of the crystal. This stone has been heat treated to darken the stone and enhance visibility of the star.
| Mineral Information |
|
[1] Minerals on Geology.com: Links to content about minerals on the Geology.com website.
[2] USGS National Minerals Information Center: Links to information about minerals on the United States Geological Survey website. [3] State and National Geological Surveys: Links to geological survey websites, most of which contain information about minerals found and produced in their location. [4] USGS Mineral Resources Program: The sole federal source of scientific information on mineral potential, production, consumption, and environmental effects. [5] The Mineral and Locality Database: The largest mineralogy database and reference website on the internet. |
Importance of Physical Properties
The primary characteristics of a mineral that determine its physical properties are its composition and the strength of the bonds in its ordered internal structure. Here are some examples:
Galena, a lead sulfide, has a much higher specific gravity than bauxite, an aluminum hydroxide. This difference is because of their composition. Lead is much heavier than aluminum.
Diamond and graphite both consist of pure carbon. Diamond is the hardest natural mineral, and graphite is one of the softest. This difference occurs because of the types of bonds connecting the carbon atoms in their mineral structures. Each carbon atom in diamond is bonded to four other carbon atoms with strong covalent bonds. Graphite has a sheet structure in which atoms within the sheets are bonded to one another with strong covalent bonds, but the bonds between the sheets are weak electrical bonds. When graphite is scratched the weak bonds fail easily, making it a soft mineral.
The gemstones ruby and sapphire are color variations of the mineral corundum. These color differences are caused by composition. When corundum contains trace amounts of chromium, it exhibits the red color of a ruby. However, when it contains trace amounts of iron or titanium, it exhibits the blue color of sapphire. If, at the time of crystallization, enough titanium is present to form tiny crystals of the mineral rutile, a star sapphire may form. This occurs when tiny crystals of rutile align systematically within the crystalline structure of the corundum to give it a silky luster that might produce a "star" that aligns with the primary crystallographic axis (see photo).
| More Minerals |
 |
Herkimer Diamonds |
 |
The Acid Test |
 |
Tumbled Stones |
 |
Zircon |
 |
Fool*s Gold |
 |
Kyanite |
 |
Rock Tumblers |
 |
Rhodochrosite |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
