Garnet
Best known as a red gemstone and birthstone of January.
Garnet occurs in many colors and has many industrial uses.
Author: Hobart M. King, PhD, GIA Graduate Gemologist

Gem garnets: Most people think that garnet is a red gemstone. However, garnet occurs in a wide variety of colors. Clockwise from the top left: red almandine (Madagascar), green tsavorite (Tanzania), yellow mali (Mali), orange spessartite (Mozambique), pink malaya (Tanzania), green merelani mint (Tanzania), red pyrope (Cote d'Ivoire), green demantoid (Namibia), purple rhodolite (Mozambique), and orange hessonite (Sri Lanka). Nine out of ten of the garnets above are from Africa, the relatively new source of spectacular garnets.
What is Garnet?
Garnet is the name used for a large group of rock-forming minerals. These minerals share a common crystal structure and a generalized chemical composition of X3Y2(SiO4)3. In that composition, "X" can be Ca, Mg, Fe2+ or Mn2+, and "Y" can be Al, Fe3+, Mn3+, V3+ or Cr3+.
These minerals are found throughout the world in metamorphic, igneous, and sedimentary rocks. Most garnet found near Earth's surface forms when a sedimentary rock with a high aluminum content, such as shale, is subjected to heat and pressure intense enough to produce schist or gneiss. Garnet is also found in the rocks of contact metamorphism, subsurface magma chambers, lava flows, deep-source volcanic eruptions, and the soils and sediments formed when garnet-bearing rocks are weathered and eroded.
Most people associate the word "garnet" with a red gemstone; however, they are often surprised to learn that garnet occurs in many other colors and has many other uses. In the United States, the major industrial uses of garnet in 2012 were waterjet cutting (35%), abrasive blasting media (30%), water filtration granules (20%), and abrasive powders (10%).
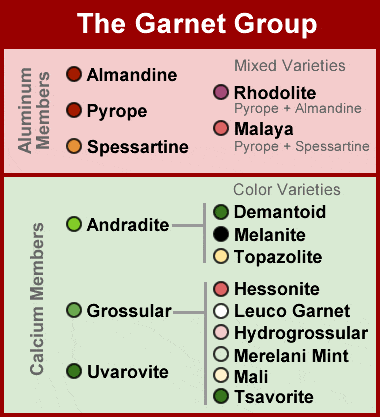
The Garnet Group: This chart summarizes the members of the garnet group that are most important as gemstones. The aluminum garnets are normally red in color with a higher specific gravity and hardness. The calcium members are usually green in color and have a lower hardness.
Physical Properties of Garnet |
|
| Chemical Classification | Silicate |
| Color | Typically red, but can be orange, green, yellow, purple, black, or brown. Blue garnets are extremely rare. |
| Streak | Colorless |
| Luster | Vitreous |
| Diaphaneity | Transparent to translucent |
| Cleavage | None |
| Mohs Hardness | 6.5 to 7.5 |
| Specific Gravity | 3.5 to 4.3 |
| Diagnostic Properties | Hardness, specific gravity, isometric crystal form, lack of cleavage |
| Chemical Composition | General formula: X3Y2(SiO4)3 |
| Crystal System | Isometric |
| Uses | Waterjet cutting granules, abrasive blasting granules, filtration granules, abrasive grits and powders, gemstones |
Garnet Physical and Chemical Properties
The most commonly encountered minerals in the garnet group include almandine, pyrope, spessartine, andradite, grossular, and uvarovite. They all have a vitreous luster, a transparent-to-translucent diaphaneity, a brittle tenacity, and a lack of cleavage. They can be found as individual crystals, stream-worn pebbles, granular aggregates, and massive occurrences. Their chemical composition, specific gravity, hardness, and colors are listed below.
Garnet Minerals |
||||
| Mineral | Composition | Specific Gravity | Hardness | Colors |
| Almandine | Fe3Al2(SiO4)3 | 4.20 | 7 - 7.5 | red, brown |
| Pyrope | Mg3Al2(SiO4)3 | 3.56 | 7 - 7.5 | red to purple |
| Spessartine | Mn3Al2(SiO4)3 | 4.18 | 6.5 - 7.5 | orange to red to brown |
| Andradite | Ca3Fe2(SiO4)3 | 3.90 | 6.5 - 7 | green, yellow, black |
| Grossular | Ca3Al2(SiO4)3 | 3.57 | 6.5 - 7.5 | green, yellow, red, pink, clear |
| Uvarovite | Ca3Cr2(SiO4)3 | 3.85 | 6.5 - 7 | green |
| The compositions listed above are for end members of several solid solution series. There are a number of other garnet minerals that are less frequently encountered and not as important in industrial use. They include goldmanite, kimzeyite, morimotoite, schorlomite, hydrogrossular, hibschite, katoite, knorringite, majorite, and calderite. | ||||
As seen above, there are a variety of different types of garnet, and each has a different chemical composition. There are also solid solution series between most of the garnet minerals. This wide variation in chemistry determines many of their physical properties. As an example, the calcium garnets generally have a lower specific gravity, a lower hardness and are typically green in color. In contrast, the iron and manganese garnets have a higher specific gravity, a greater hardness and are typically red in color.

Almandine garnet: Excellent cubic crystals of almandine garnet in a fine-grained mica schist from Granatenkogel Mountain, Austria. Specimen and photo by Arkenstone / www.iRocks.com.

Andradite garnet: Green andradite garnet of the demantoid variety on a matrix of marble. This specimen is about 8.9 x 6.5 x 4.8 centimeters in size and was collected in Antsiranana Province, Madagascar. Garnets formed within marble often have excellent crystal form and are of very high quality. Specimen and photo by Arkenstone / www.iRocks.com.

Garnet gneiss: A coarse-grained gneiss composed mainly of hornblende (black), plagioclase (white) and garnet (red) from Norway. Public domain photo by Woudloper.
How Does Garnet Form?
Garnet in Metamorphic Rocks
Most garnet forms at convergent plate boundaries where shale is being acted upon by regional metamorphism. The heat and pressure of metamorphism breaks chemical bonds and causes minerals to recrystallize into structures that are stable under the new temperature-pressure environment. The aluminum garnet, almandine, generally forms in this environment.
As these rocks are metamorphosed, the garnets start as tiny grains and enlarge slowly over time as metamorphism progresses. As they grow, they displace, replace, and include the surrounding rock materials. The photo below shows a microscopic view of a garnet grain that has grown within a schist matrix. It included a number of the host rock's mineral grains as it grew. This explains why so many garnets formed by regional metamorphism are highly included.

Garnet mica schist in thin section: This is a microscopic view of a garnet grain that has grown in schist. The large black grain is the garnet, the red elongate grains are mica flakes. The black, gray, and white grains are mostly silt or smaller size grains of quartz and feldspar. The garnet has grown by replacing, displacing, and including the mineral grains of the surrounding rock. You can see many of these grains as inclusions within the garnet. From this photo it is easy to understand why clean, gem-quality garnets with no inclusions are very hard to find. It is also hard to understand how garnet can grow into nice euhedral crystals under these conditions. Photo by Jackdann88, displayed here under a Creative Commons license.
The calcium garnets typically form when argillaceous limestone is altered into marble by contact metamorphism along the edges of igneous intrusions. These are andradite, grossular, and uvarovite, the slightly softer, typically green garnets with a lower specific gravity. Two calcium garnets are highly regarded in the gem trade; they are tsavorite (a bright green grossular) and demantoid (a golden-green andradite).
Garnet is a common mineral in skarns. Skarns are metamorphic rocks that have been chemically and mineralogically altered by metasomatism. In the metasomatism process, hot, chemically active fluids flow through the rocks to cause recrystallization and compositional change. Skarns are usually rich in garnet, pyroxene minerals, carbonate minerals, and quartz.
Garnet often occurs as an accessory mineral in igneous rocks such as granite. Many people are familiar with almandine garnet because it is sometimes seen as dark red crystals in the igneous rocks used as granite countertops. Spessartine is an orange garnet found as crystals in granite pegmatites. Pyrope is a red garnet that is brought to Earth's surface in pieces of peridotite that were torn from the mantle during deep-source volcanic eruptions. Garnet is also found in basaltic lava flows.

Alluvial garnet crystals: These almandine-spessartine garnets are from an alluvial deposit in Idaho. They have been transported a short distance from their source rock, and some still retain evidence of their dodecahedral crystal form. They are about four to five millimeters in size and weigh about 0.6 to 0.8 carats each.
Garnet in Sedimentary Rocks and Sediments
Garnets are relatively durable minerals. They are often found concentrated in the soils and sediments that form when garnet-bearing rocks are weathered and eroded. These alluvial garnets are often the target of mining operations because they are easy to mine and remove from the sediment/soil by mechanical processing.
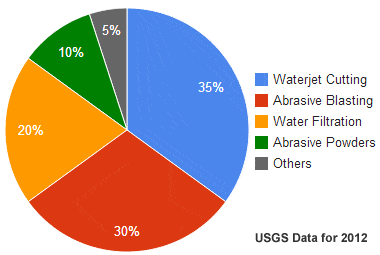
Uses of Garnet: This chart shows the most common industrial uses of garnet minerals. Almandine is the variety of garnet that is most often used in industry.
Uses of Garnet
Garnet has been used as a gemstone for thousands of years. In the past 150 years, it has seen many additional uses as an industrial mineral. The chart below shows recent industrial uses of garnet in the United States. Garnet is also used as an indicator mineral during mineral exploration and geologic assessments.

Garnet abrasive: This photo shows garnet granules that have been crushed and size-graded for use as abrasive, cutting, and filter media. They are used in waterjet cutting, "sand" blasting, sandpaper, water filtration, and a number of other uses. Almandine is the hardest garnet and also the most abundant. It is the garnet of choice for most abrasive applications. Photo by the United States Geological Survey.
Garnet as an Industrial Mineral
Garnet Abrasives
The first industrial use of garnet was as an abrasive. Garnet is a relatively hard mineral with a hardness that ranges between 6.5 and 7.5 on the Mohs Scale. That allows it to be used as an effective abrasive in many types of manufacturing. When crushed, it breaks into angular pieces that provide sharp edges for cutting and sanding. Small granules of uniform size are bonded to paper to produce a reddish color sandpaper that is widely used in woodworking shops. Garnet is also crushed, screened to specific sizes, and sold as abrasive granules and powders. In the United States, New York and Idaho have been important sources of industrial garnet for abrasives.

Garnet Sandpaper: Crushed garnet granules are used to make garnet sandpaper. Garnet serves as an excellent abrasive, especially for sanding wood. The crushed garnet granules are very sharp, and as the paper is used the granules fracture to expose new sharp surfaces. If you see sandpaper covered with reddish brown granules, look at the back to see if it is garnet paper.

Garnet crystal: Almandine, a variety of garnet from River Valley, Ontario, Canada. This specimen is a nice euhedral crystal approximately 2 inches (5 centimeters) across. These types of crystals are often weathered out of a garnet-bearing mica schist and are transported by streams.
The largest industrial use of garnet in the United States is in waterjet cutting. A machine known as a waterjet cutter produces a high-pressure jet of water with entrained abrasive granules. When these are directed at a piece of metal, ceramic, or stone, a cutting action can occur that produces very little dust and cuts at a low temperature. Waterjet cutters are used in manufacturing and mining.

Almandine garnet: Almandine, a variety of garnet from Lount Township, Ontario, Canada. This is a granular specimen approximately 11.4 centimeters across.
Garnet granules are also used in abrasive blasting (commonly known as "sand blasting"). In these processes, a tool propels a stream of abrasive granules (also known as "media") against a surface using a highly pressurized fluid (usually air or water) as a propellant. Abrasive blasting is done in order to smooth, clean, or remove oxidation products from metals, brick, stone, and other materials. It is usually much faster than sanding by hand or with a sanding machine. It can clean small and intricate surfaces that other cleaning methods would miss. Abrasives of various hardnesses can be used to clean a surface of greater hardness, without damaging the surface.
Garnet granules are often used as a filter media. Small garnet particles are used to fill a container through which a liquid flows. The pore spaces of the garnet are small enough to allow passage of the liquid but are too small to allow passage of some contaminant particles, which are filtered from the flow. Garnet is suited for this use because it is relatively inert and has a relatively high specific gravity. Garnet granules, crushed and graded to about 0.3 millimeters in size, can be used to filter out contaminant particles as small as a few microns in diameter. Garnet's high specific gravity and high hardness reduce bed expansion and particle abrasion during backflushing.

Garnet peridotite: Garnet peridotite from Alpe Arami, near Bellinzona, Switzerland. The material in this rock originated within Earth's mantle and was delivered to the surface through a volcanic pipe during a deep-source volcanic eruption. The garnets are the reddish purple grains within the rock. Garnets weathered from such pipes often serve as indicator minerals when exploring for volcanic pipes that might contain diamond. Public domain photo by Woudloper.

The best way to learn about minerals is to study with a collection of small specimens that you can handle, examine, and observe their properties. Inexpensive mineral collections are available in the Geology.com Store. Image copyright iStockphoto / Anna Usova.
Garnet as a Geological Indicator Mineral
Although most of the garnets found at Earth's surface have formed within the crust, some garnets are brought up from the mantle during deep-source volcanic eruptions. These eruptions entrain pieces of mantle rock known as "xenoliths" and deliver them to the surface in a structure known as a "pipe." These xenoliths are the source of most diamonds found at or near Earth's surface.
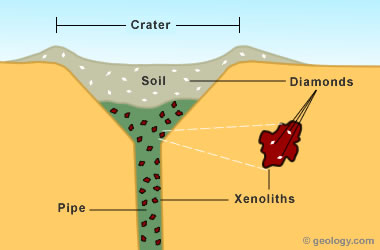
Diamond pipe: Simplified cross-section of a diamond pipe and residual soil deposit showing the relationships of xenoliths and diamonds with the pipe and residual soil.
Although xenoliths contain diamonds, they often contain a tremendous number of garnets for every diamond, and those garnets are generally larger in size. These deep-source garnets are very different from the garnets that form in the crust at shallow depth. So, a good way to prospect for diamonds is to look for these unique garnets. The garnets serve as "indicator minerals" for geologists exploring for diamond deposits. As the xenoliths weather, their garnets are liberated in large numbers. These unusual garnets then move downslope in soils and streams. Geologists who find them can follow the garnet trail to the source deposit. Some of the diamond pipes in Canada were found by following a garnet trail produced by moving ice.

African garnets: African garnets of various colors: orange spessartine (Mozambique), yellow mali (Mali), red almandine (Madagascar), green tsavorite (Tanzania), and purple rhodolite (Mozambique). In the past two decades, Africa has become a major source of excellent beautiful garnets with great color and clarity.

Melanite garnet: Melanite is an opaque black garnet that is unusual to find in jewelry today. Along with jet, black chalcedony, and other black gems, melanite was often used in jewelry during the Victorian Era. These two rose-cut melanite rounds are about 9 millimeters across.
Garnets as Gemstones
Garnet has been used as a gemstone for over 5000 years. It has been found in the jewelry of many Egyptian burials and was the most popular gemstone of Ancient Rome. It is a beautiful gem that is usually sold without treatment of any kind. It is also durable and common enough that it can be used in jewelry at a relatively low cost.
Garnet continues as a popular gemstone today. It serves as a birthstone for the month of January and is a traditional gem given on a second anniversary. Most people will think of a red gemstone when they hear the name "garnet" because they are not aware that garnet occurs in a variety of colors. However, gem-quality garnets occur in every color - with red being the most common and blue garnets being especially rare.
Red almandine is the red garnet most often found in jewelry because it is abundant and inexpensive. Pyrope and spessartine are reddish garnets that are commonly encountered in jewelry for the same reason. In recent decades, green demantoid garnet has become popular. It has a dispersion of 0.057 that gives it a "fire" which exceeds that of diamond's at 0.044. Green tsavorite has a bright, rich color that is very much like emerald. It is commonly used as an alternative stone to emerald. Both of these green garnets are becoming more popular, but their price is much higher than almandine.
| Garnet Information |
|
[1] Gemstones of the World: Walter Schumann; Sterling Publishing; fifth edition; 320 pages; 2013.
[2] Garnet -- An Essential Industrial Mineral and January's Birthstone: James G. Evans, Phillip R. Moyle, David G. Frank and Donald W. Olson; United States Geological Survey Fact Sheet 2006-3149; 2006. [3] Industrial Garnet: by Donald W. Olson; United States Geological Survey 2012 Minerals Yearbook, August 2016. [4] Industrial Garnet: by Donald W. Olson; United States Geological Survey Mineral Commodity Summaries, February 2014. |
| More Minerals |
 |
Herkimer Diamonds |
 |
The Acid Test |
 |
Tumbled Stones |
 |
Zircon |
 |
Fool*s Gold |
 |
Kyanite |
 |
Rock Tumblers |
 |
Rhodochrosite |

Find Other Topics on Geology.com:

|

| ||

|

| ||

|

| ||

|

|
